UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 6-K
REPORT OF FOREIGN PRIVATE ISSUER PURSUANT TO RULE 13a‑16 OR 15d‑16
UNDER THE SECURITIES EXCHANGE ACT OF 1934
FOR THE MONTH OF JANUARY 2026
COMMISSION FILE NUMBER 001-39081
BioNTech SE
(Translation of registrant’s name into English)
(Translation of registrant’s name into English)
An der Goldgrube 12
D-55131 Mainz
Germany
+49 6131-9084-0
(Address of principal executive offices)
(Address of principal executive offices)
Indicate by check mark whether the registrant files or will file annual reports under cover Form 20‑F or Form 40‑F: Form 20‑F ☒ Form 40‑F ☐
Indicate by check mark if the registrant is submitting the Form 6‑K in paper as permitted by Regulation S‑T Rule 101(b)(1): ☐
Indicate by check mark if the registrant is submitting the Form 6‑K in paper as permitted by Regulation S‑T Rule 101(b)(7): ☐
DOCUMENTS INCLUDED AS PART OF THIS FORM 6-K
On January 13, 2026, BioNTech SE provided a strategic business update and outlined the Company’s focus areas for 2026, including an overview of expected near- to longer-term milestones, at the 44th Annual J.P. Morgan Healthcare Conference in San Francisco, California. The presentation is attached hereto as Exhibit 99.1.
SIGNATURE
Pursuant to the requirements of the Exchange Act, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| BioNTech SE | ||||||||||||||
| By: | /s/ Ramon Zapata-Gomez | By: | /s/ Dr. Sierk Poetting | |||||||||||
| Name: Ramon Zapata-Gomez | Name: Dr. Sierk Poetting | |||||||||||||
| Title: Chief Financial Officer | Title: Chief Operating Officer | |||||||||||||
Date: January 13, 2026
EXHIBIT INDEX
| Exhibit | Description of Exhibit | ||||
| 99.1 | |||||

Translating Science into Survival Prof. Dr. Ugur Sahin, M.D., CEO & Co-founder, BioNTech 44th J.P. Morgan Healthcare Conference January 13, 2026 Exhibit 99.1

2 This Slide Presentation Includes Forward-Looking Statements This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, as amended, including, but not limited to, statements concerning: BioNTech’s expected revenues and net profit/(loss) related to sales of BioNTech’s COVID-19 vaccine, referred to as COMIRNATY where approved for use under full or conditional marketing authorization, in territories controlled by BioNTech’s collaboration partners, particularly for those figures that are derived from preliminary estimates provided by BioNTech’s partners; the rate and degree of market acceptance of BioNTech’s COVID-19 vaccine and, if approved, BioNTech’s investigational medicines; the initiation, timing, progress, results, and cost of BioNTech’s research and development programs, including BioNTech’s current and future preclinical studies and clinical trials, including statements regarding the expected timing of initiation, enrollment, and completion of studies or trials and related preparatory work and the availability of results, and the timing and outcome of applications for regulatory approvals and marketing authorizations; BioNTech’s expectations regarding potential future commercialization in oncology, including goals regarding timing and indications; the targeted timing and number of additional potentially registrational trials, and the registrational potential of any trial BioNTech may initiate; discussions with regulatory agencies; BioNTech’s expectations with respect to intellectual property; the impact of BioNTech’s collaboration and licensing agreements, including BioNTech’s partnership with BMS; the development, nature and feasibility of sustainable vaccine production and supply solutions; the deployment of AI across BioNTech’s preclinical and clinical operations; BioNTech's expectations for upcoming scientific presentations; and BioNTech’s expectations of net profit / (loss). In some cases, forward-looking statements can be identified by terminology such as “will,” “may,” “should,” “expects,” “intends,” “plans,” “aims,” “anticipates,” “believes,” “estimates,” “predicts,” “potential,” “continue,” or the negative of these terms or other comparable terminology, although not all forward-looking statements contain these words. The forward-looking statements in this presentation are based on BioNTech’s current expectations and beliefs of future events and are neither promises nor guarantees. You should not place undue reliance on these forward- looking statements because they involve known and unknown risks, uncertainties, and other factors, many of which are beyond BioNTech’s control, and which could cause actual results to differ materially and adversely from those expressed or implied by these forward-looking statements. These risks and uncertainties include, but are not limited to: the uncertainties inherent in research and development, including the ability to meet anticipated clinical endpoints, commencement and/or completion dates for clinical trials, projected data release timelines, regulatory submission dates, regulatory approval dates and/or launch dates, as well as risks associated with preclinical and clinical data, including the data discussed in this presentation, and including the possibility of unfavorable new preclinical, clinical or safety data and further analyses of existing preclinical, clinical or safety data; the nature of the clinical data, which is subject to ongoing peer review, regulatory review and market interpretation; BioNTech’s pricing and coverage negotiations regarding its COVID-19 vaccine with governmental authorities, private health insurers and other third-party payors; the future commercial demand and medical need for initial or booster doses of a COVID-19 vaccine; the impact of tariffs and escalations in trade policy; competition from other COVID-19 vaccines or related to BioNTech’s other product candidates, including those with different mechanisms of action and different manufacturing and distribution constraints, on the basis of, among other things, efficacy, cost, convenience of storage and distribution, breadth of approved use, side-effect profile and durability of immune response; the timing of and BioNTech’s ability to obtain and maintain regulatory approval for its product candidates; the ability of BioNTech’s COVID-19 vaccines to prevent COVID-19 caused by emerging virus variants; BioNTech’s and its counterparties’ ability to manage and source necessary energy resources; BioNTech’s ability to identify research opportunities and discover and develop investigational medicines; the ability and willingness of BioNTech’s third-party collaborators to continue research and development activities relating to BioNTech's development candidates and investigational medicines; the impact of COVID-19 on BioNTech’s development programs, supply chain, collaborators and financial performance; unforeseen safety issues and potential claims that are alleged to arise from the use of products and product candidates developed or manufactured by BioNTech; BioNTech’s and its collaborators’ ability to commercialize and market BioNTech’s COVID-19 vaccine and, if approved, its product candidates; BioNTech’s ability to manage its development and related expenses; regulatory and political developments in the United States and other countries; BioNTech’s ability to effectively scale its production capabilities and manufacture its products and product candidates; risks relating to the global financial system and markets; and other factors not known to BioNTech at this time. You should review the risks and uncertainties described under the heading “Risk Factors” in BioNTech’s Report on Form 6-K for the period ended September 30, 2025, and in subsequent filings made by BioNTech with the SEC, which are available on the SEC’s website at www.sec.gov. These forward-looking statements speak only as of the date hereof. Except as required by law, BioNTech disclaims any intention or responsibility for updating or revising any forward-looking statements contained in this presentation in the event of new information, future developments or otherwise. Furthermore, certain statements contained in this presentation relate to or are based on studies, publications, surveys and other data obtained from third-party sources and BioNTech’s own internal estimates and research. While BioNTech believes these third-party sources to be reliable as of the date of this presentation, it has not independently verified, and makes no representation as to the adequacy, fairness, accuracy or completeness of, any information obtained from third-party sources. In addition, any market data included in this presentation involves assumptions and limitations, and there can be no guarantee as to the accuracy or reliability of such assumptions. While BioNTech believes its own internal research is reliable, such research has not been verified by any independent source. In addition, BioNTech is the owner of various trademarks, trade names and service marks that may appear in this presentation. Certain other trademarks, trade names and service marks appearing in this presentation are the property of third parties. Solely for convenience, the trademarks and trade names in this presentation may be referred to without the ® and TM symbols, but such references should not be construed as any indicator that their respective owners will not assert, to the fullest extent under applicable law, their rights thereto. An abbreviation directory of defined terms can be found at the end of the presentation.

COVID-19 Vaccine Global Impact Addressing Oncology Unmet Medical Need Focused Infectious Disease Innovation 5 Billion vaccine doses distributed1 >25 Ongoing Phase 2 & Phase 3 trials2 16 Clinical programs3 6 High unmet need clinical programs4 Continuing to Execute on BioNTech’s Strategy 3 1. Includes globally distributed doses from 2020 to-date; 2. Includes Phase 2 or 3 trials for BNT111, BNT113, autogene cevumeran, gotistobart, trastuzumab pamirtecan and pumitamig; 3. Includes BNT111, BNT113, BNT116, autogene cevumeran, BNT211, BNT314/GEN1059, gotistobart, BNT317, trastuzumab pamirtecan, BNT324/DB-1311, BNT325/DB-1305, BNT326/YL202, pumitamig, BNT329, BNT3212, BNT3213; 4. Includes BNT162, BNT161, BNT163, BNT164. BNT165, BNT166. In-House GMP Manufacturing Platforms Fully Integrated AI-Driven Innovation Capabilities and facilities for key platforms: mRNA therapeutics, including individualized mRNA, and bispecific antibodies Tech-bio company with AI-infused target and drug discovery and development capabilities

2025 Achievements: Strong Performance and Pipeline Momentum 4 COVID-19 Market Leadership Advanced Key Oncology Programs Executed Key Strategic Deals Strengthened Financial Position Launched variant-adapted COVID-19 vaccine Leading COVID-19 vaccine market share1 Over 25 phase 2 & 3 oncology trials ongoing2 10 novel-combination trials ongoing with pumitamig3 Strategic BMS partnership Acquired Biotheus4 Acquired CureVac5 Increased 2025 revenue guidance6 €17.2 billion in cash, cash equivalents and securities7 1. Over 50%, including Italy, Spain, France, Germany, USA, Japan, Australia; 2. Includes Phase 2 or 3 trials for BNT111, BNT113, autogene cevumeran (partnered with Genentech, a member of the Roche Group), gotistobart (partnered with OncoC4), trastuzumab pamirtecan (partnered with DualityBio) and pumitamig (partnered with Bristol Myers Squibb) 3. Partnered with Bristol Myers Squibb (BMS); 4. Close announced on February 4, 2025; 5. Close announced on January 6, 2026; 6. BioNTech increased revenue guidance on November 3, 2025 and now expects its revenues for the full 2025 financial year to be in the range of €2,600 - €2,800 million, from previous range of €1,700 - €2,200 million; please refer to 3Q25 earnings press release and quarterly report on Form 6-K for risks and uncertainties. 7. Preliminary, unaudited figure; consists of cash, cash equivalents and security investments, as of December 31, 2025.
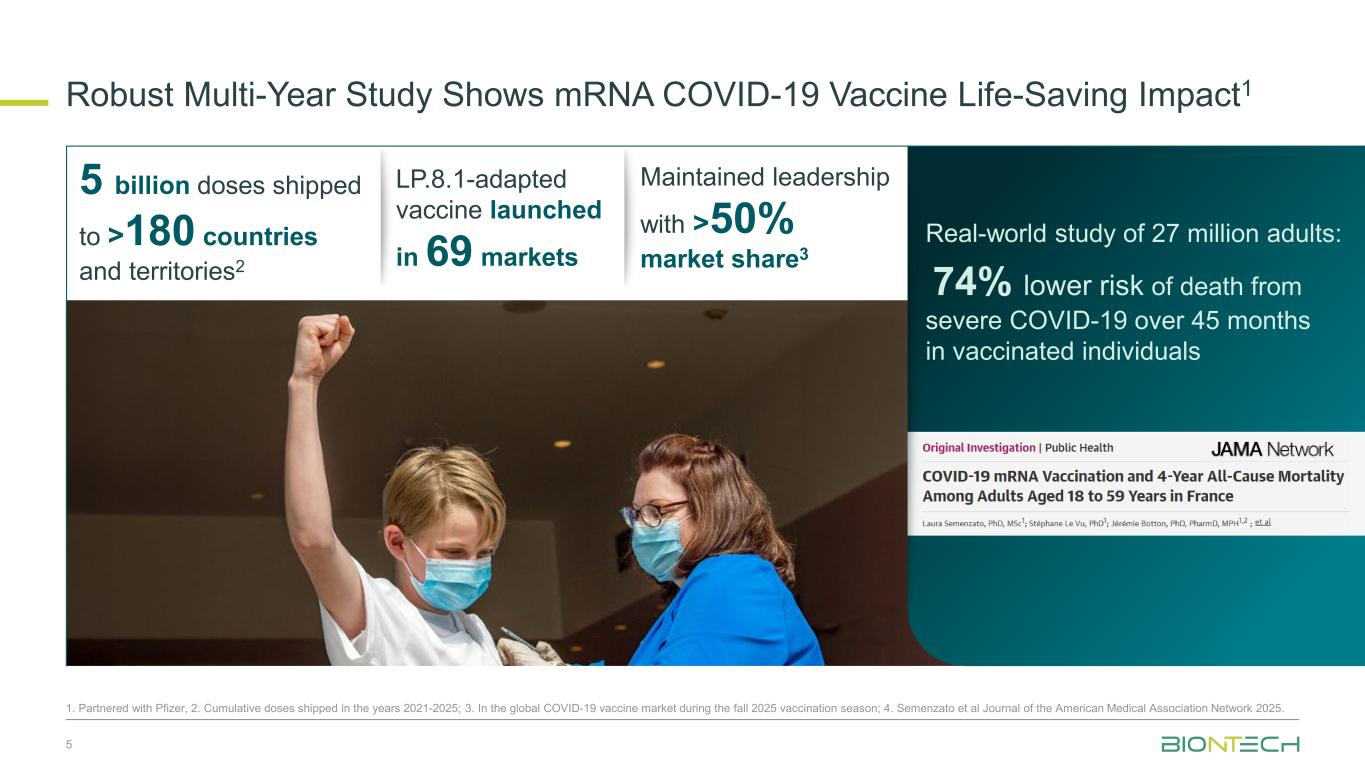
Robust Multi-Year Study Shows mRNA COVID-19 Vaccine Life-Saving Impact1 5 1. Partnered with Pfizer, 2. Cumulative doses shipped in the years 2021-2025; 3. In the global COVID-19 vaccine market during the fall 2025 vaccination season; 4. Semenzato et al Journal of the American Medical Association Network 2025. Real-world study of 27 million adults: 74% lower risk of death from severe COVID-19 over 45 months in vaccinated individuals 5 billion doses shipped to >180 countries and territories2 LP.8.1-adapted vaccine launched in 69 markets Maintained leadership with >50% market share3
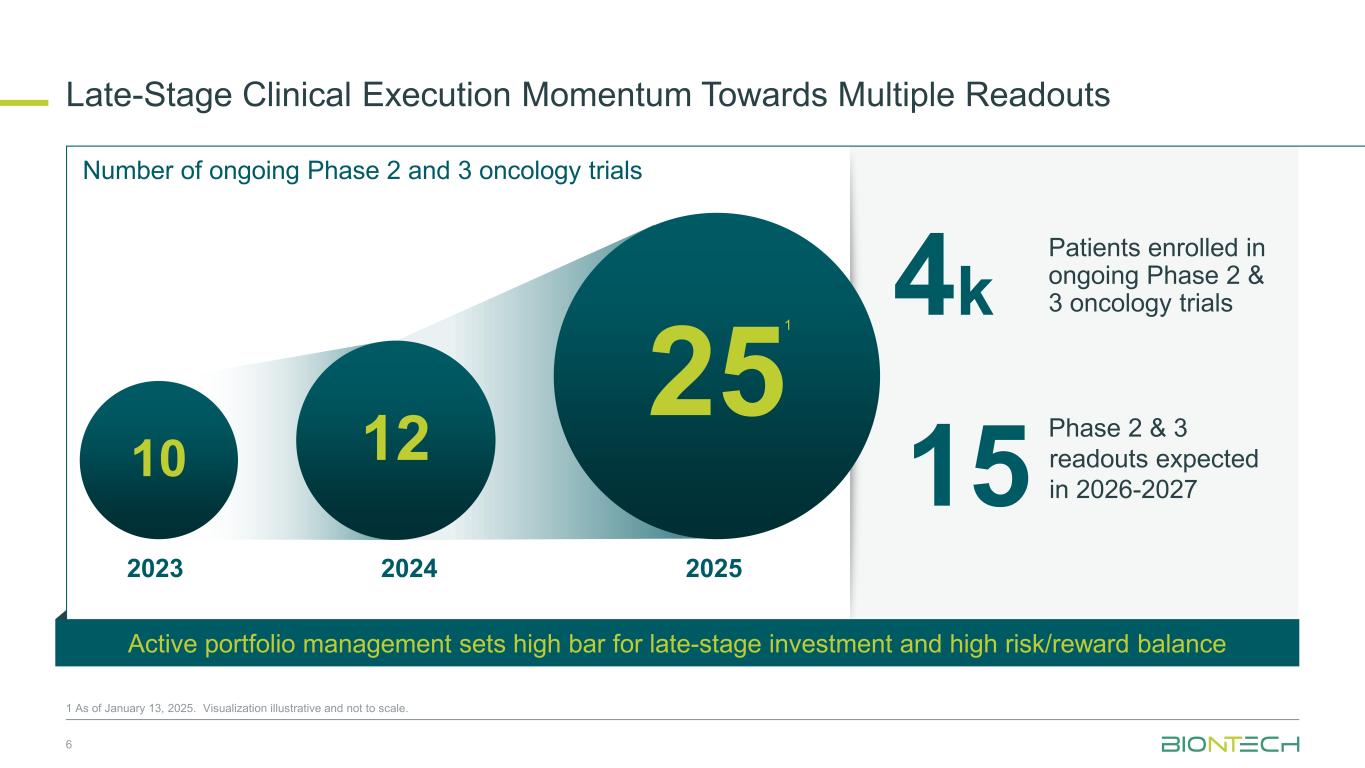
Late-Stage Clinical Execution Momentum Towards Multiple Readouts 6 1 As of January 13, 2025. Visualization illustrative and not to scale. 4k Number of ongoing Phase 2 and 3 oncology trials 1210 251 202520242023 15 Patients enrolled in ongoing Phase 2 & 3 oncology trials Phase 2 & 3 readouts expected in 2026-2027 Active portfolio management sets high bar for late-stage investment and high risk/reward balance

Strong Financial Position Drives Sustainable Oncology Innovation 7 In 2026, BioNTech anticipates a modest decline in Comirnaty revenues compared to 2025, reflecting COVID-19 vaccine market dynamics, which are influenced by various factors, including but not limited to changing vaccine recommendations, specifically in the United States, and the continued transition from multi-year contracts to private markets in different geographies. BioNTech does not currently anticipate the recognition of revenues from the sale of any oncology products in 2026. Per the outlined partnership terms, revenues to BioNTech from the collaboration with Bristol Myers Squibb in 2026 are expected to be broadly in line with 2025. 1. Preliminary, unaudited figure; consists of cash, cash equivalents and security investments, as of December 31, 2025. High-margin cash- generative COVID-19 vaccine business €17.2 billion cash balance1 de-risks oncology execution Active portfolio management focused on late- stage programs COVID-19 Vaccine Revenue Cash Balance Resource Allocation Strengthened P&L through profit- and cost-sharing Financial Strength
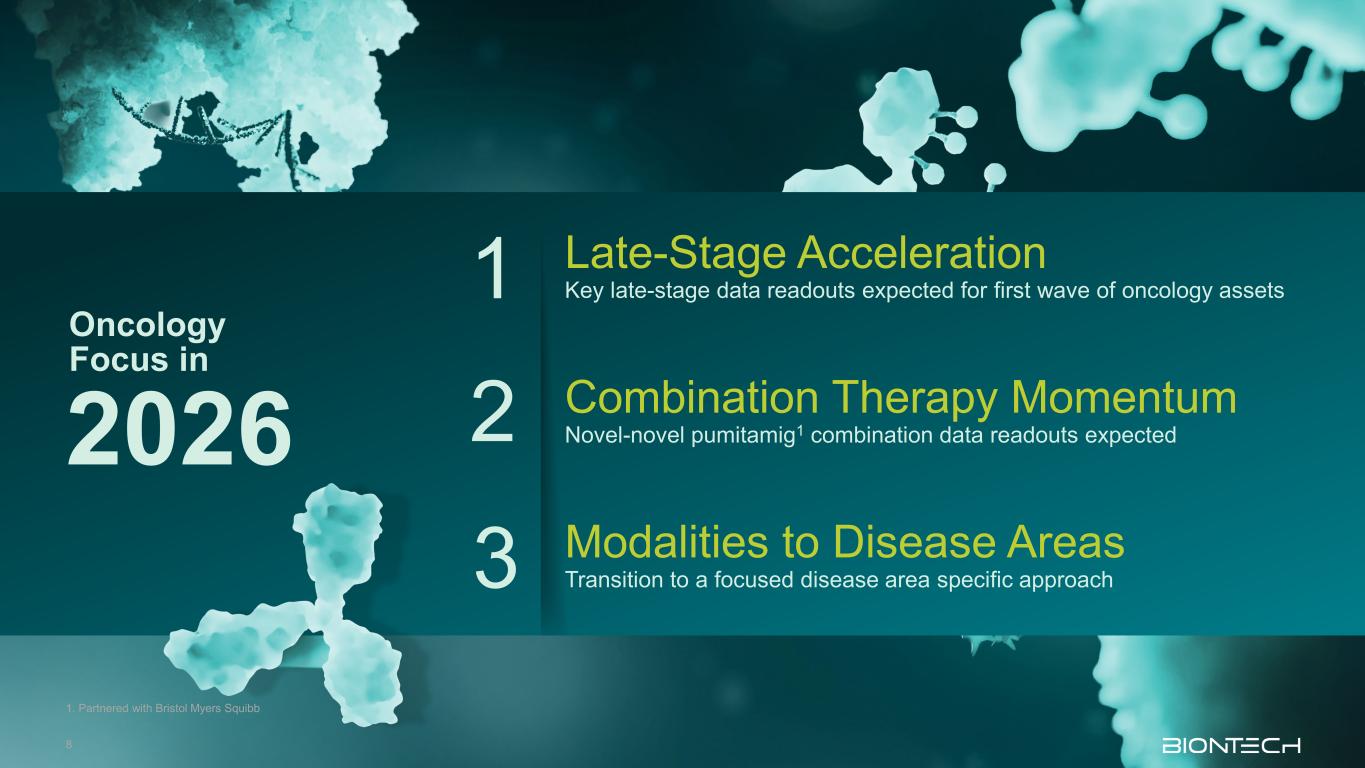
Modalities to Disease Areas Transition to a focused disease area specific approach Late-Stage Acceleration Key late-stage data readouts expected for first wave of oncology assets Oncology Focus in 2026 Combination Therapy Momentum Novel-novel pumitamig1 combination data readouts expected 1 2 3 1. Partnered with Bristol Myers Squibb 8
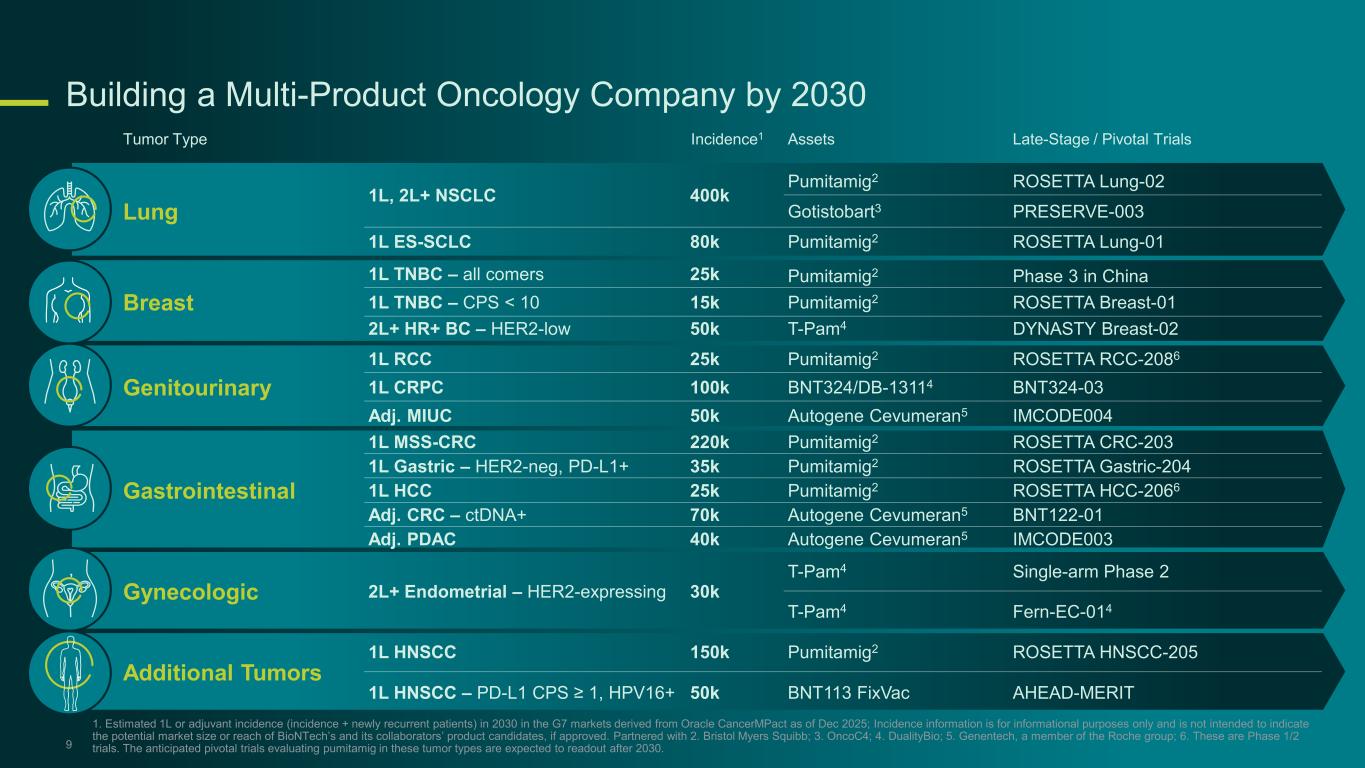
Building a Multi-Product Oncology Company by 2030 9 Tumor Type Incidence1 Assets Late-Stage / Pivotal Trials Lung 1L, 2L+ NSCLC 400k Pumitamig2 ROSETTA Lung-02 Gotistobart3 PRESERVE-003 1L ES-SCLC 80k Pumitamig2 ROSETTA Lung-01 Breast 1L TNBC – all comers 25k Pumitamig2 Phase 3 in China 1L TNBC – CPS < 10 15k Pumitamig2 ROSETTA Breast-01 2L+ HR+ BC – HER2-low 50k T-Pam4 DYNASTY Breast-02 Genitourinary 1L RCC 25k Pumitamig2 ROSETTA RCC-2086 1L CRPC 100k BNT324/DB-13114 BNT324-03 Adj. MIUC 50k Autogene Cevumeran5 IMCODE004 Gastrointestinal 1L MSS-CRC 220k Pumitamig2 ROSETTA CRC-203 1L Gastric – HER2-neg, PD-L1+ 35k Pumitamig2 ROSETTA Gastric-204 1L HCC 25k Pumitamig2 ROSETTA HCC-2066 Adj. CRC – ctDNA+ 70k Autogene Cevumeran5 BNT122-01 Adj. PDAC 40k Autogene Cevumeran5 IMCODE003 Gynecologic 2L+ Endometrial – HER2-expressing 30k T-Pam4 Single-arm Phase 2 T-Pam4 Fern-EC-014 Additional Tumors 1L HNSCC 150k Pumitamig2 ROSETTA HNSCC-205 1L HNSCC – PD-L1 CPS ≥ 1, HPV16+ 50k BNT113 FixVac AHEAD-MERIT 1. Estimated 1L or adjuvant incidence (incidence + newly recurrent patients) in 2030 in the G7 markets derived from Oracle CancerMPact as of Dec 2025; Incidence information is for informational purposes only and is not intended to indicate the potential market size or reach of BioNTech’s and its collaborators’ product candidates, if approved. Partnered with 2. Bristol Myers Squibb; 3. OncoC4; 4. DualityBio; 5. Genentech, a member of the Roche group; 6. These are Phase 1/2 trials. The anticipated pivotal trials evaluating pumitamig in these tumor types are expected to readout after 2030.
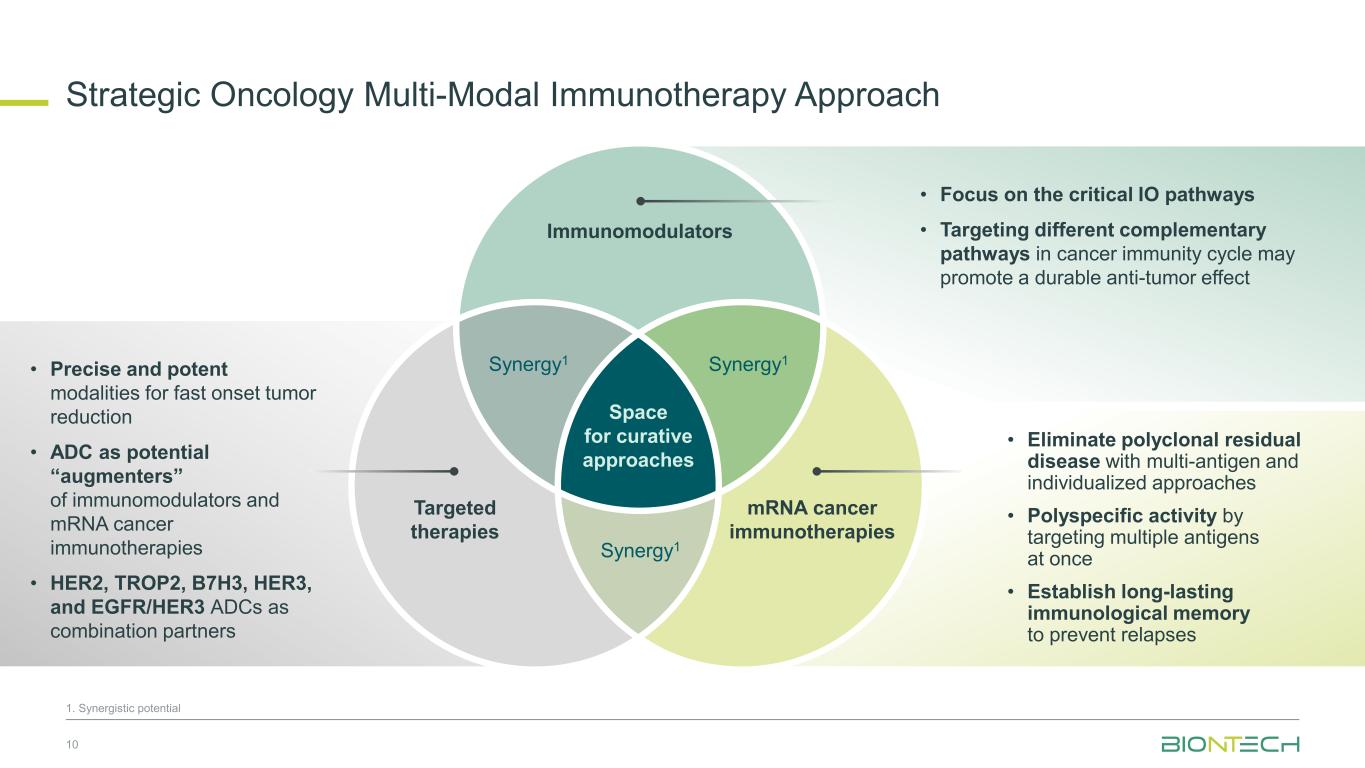
Strategic Oncology Multi-Modal Immunotherapy Approach 10 1. Synergistic potential Space for curative approaches Immunomodulators Targeted therapies Synergy1Synergy1 Synergy1 mRNA cancer immunotherapies • Focus on the critical IO pathways • Targeting different complementary pathways in cancer immunity cycle may promote a durable anti-tumor effect • Eliminate polyclonal residual disease with multi-antigen and individualized approaches • Polyspecific activity by targeting multiple antigens at once • Establish long-lasting immunological memory to prevent relapses • Precise and potent modalities for fast onset tumor reduction • ADC as potential “augmenters” of immunomodulators and mRNA cancer immunotherapies • HER2, TROP2, B7H3, HER3, and EGFR/HER3 ADCs as combination partners
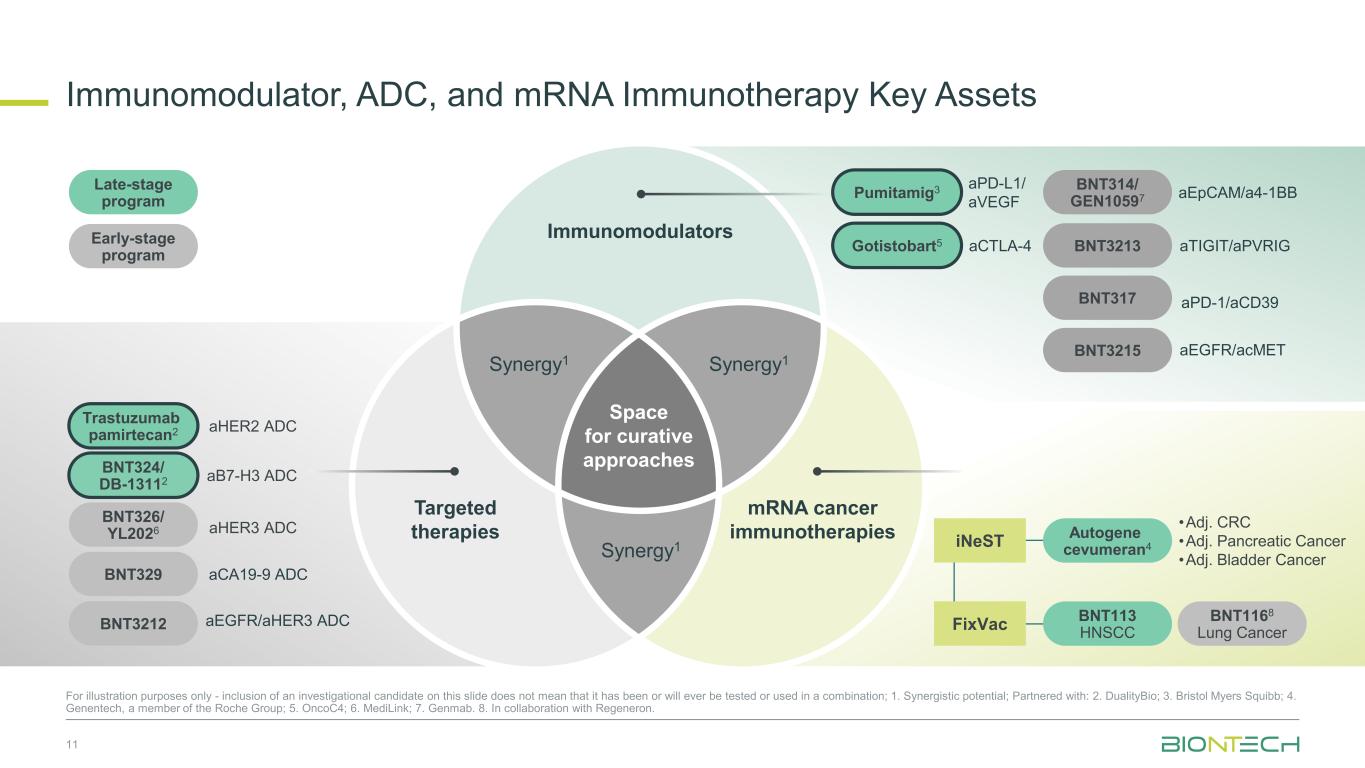
Immunomodulators Targeted therapies Synergy1Synergy1 Synergy1 mRNA cancer immunotherapies Space for curative approaches Immunomodulator, ADC, and mRNA Immunotherapy Key Assets 11 For illustration purposes only - inclusion of an investigational candidate on this slide does not mean that it has been or will ever be tested or used in a combination; 1. Synergistic potential; Partnered with: 2. DualityBio; 3. Bristol Myers Squibb; 4. Genentech, a member of the Roche Group; 5. OncoC4; 6. MediLink; 7. Genmab. 8. In collaboration with Regeneron. Trastuzumab pamirtecan2 aHER2 ADC BNT324/ DB-13112 aB7-H3 ADC BNT326/ YL2026 aHER3 ADC Gotistobart5 Pumitamig3 aCTLA-4 aPD-L1/ aVEGF iNeST FixVac BNT113 HNSCC BNT1168 Lung Cancer Autogene cevumeran4 •Adj. CRC •Adj. Pancreatic Cancer •Adj. Bladder Cancer BNT314/ GEN10597 BNT3213 BNT317 BNT3215 aPD-1/aCD39 aEpCAM/a4-1BB aTIGIT/aPVRIG aEGFR/acMET BNT329 aCA19-9 ADC BNT3212 aEGFR/aHER3 ADC Late-stage program Early-stage program
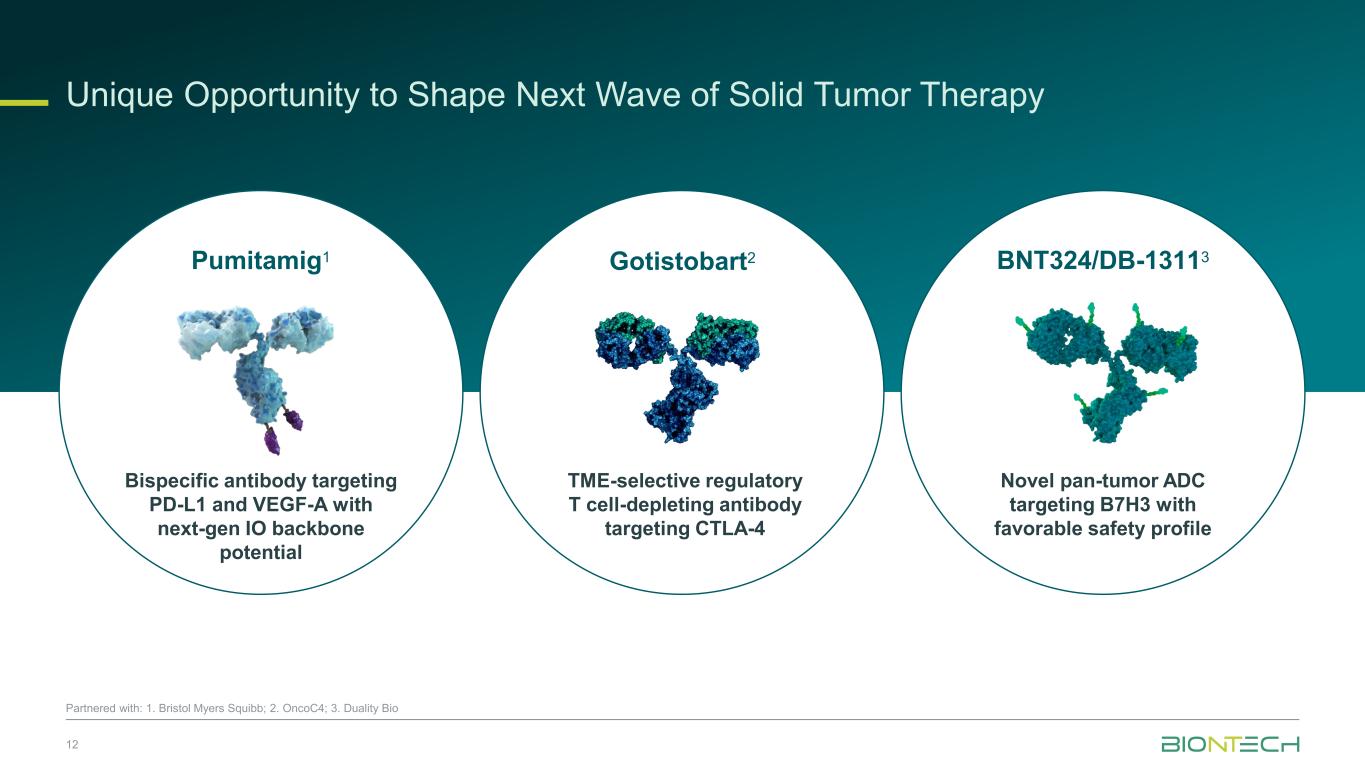
Pumitamig1 Bispecific antibody targeting PD-L1 and VEGF-A with next-gen IO backbone potential Gotistobart2 TME-selective regulatory T cell-depleting antibody targeting CTLA-4 BNT324/DB-13113 Novel pan-tumor ADC targeting B7H3 with favorable safety profile Unique Opportunity to Shape Next Wave of Solid Tumor Therapy 12 Partnered with: 1. Bristol Myers Squibb; 2. OncoC4; 3. Duality Bio
Pumitamig1 Bispecific antibody targeting PD-L1 and VEGF-A with next-gen IO backbone potential Unique Opportunity to Shape Next Wave of Solid Tumor Therapy 13 Partnered with: 1. Bristol Myers Squibb; 2. OncoC4; 3. Duality Bio
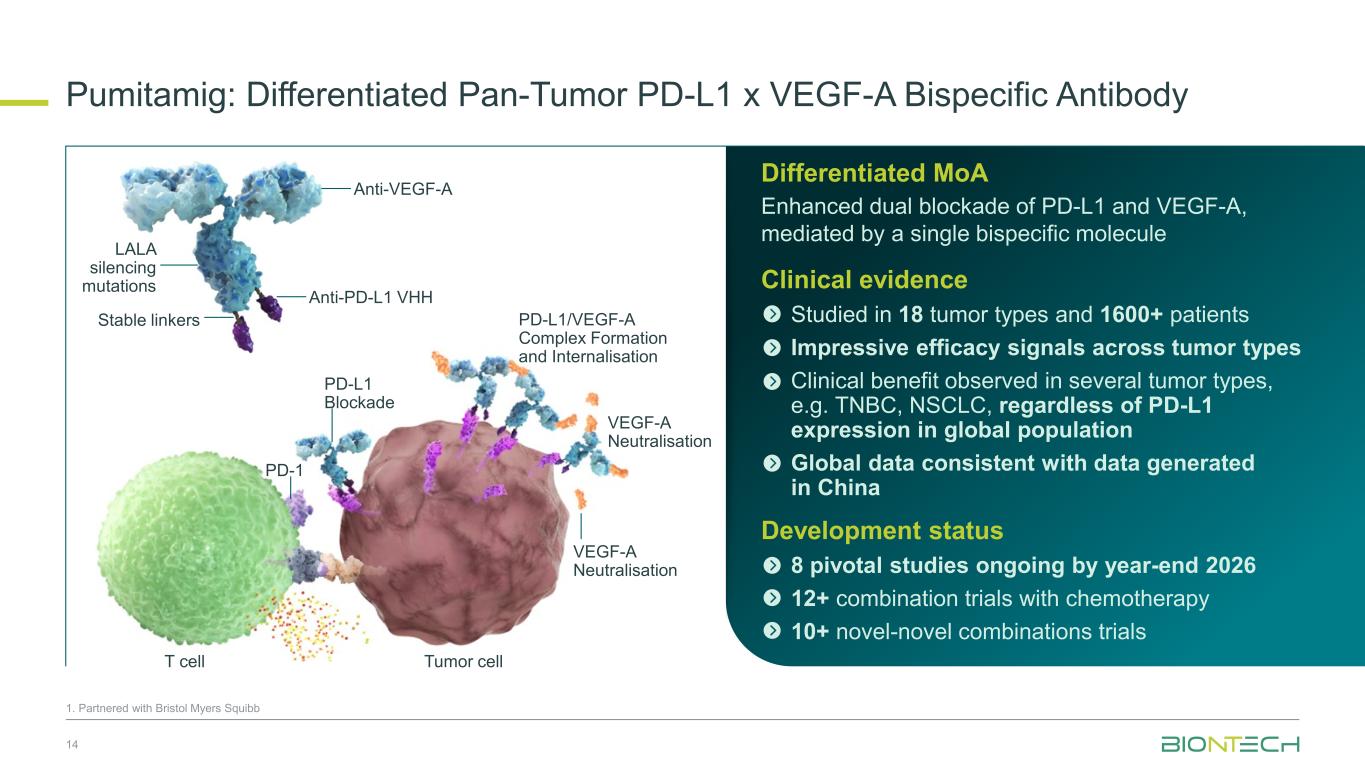
Pumitamig: Differentiated Pan-Tumor PD-L1 x VEGF-A Bispecific Antibody 14 1. Partnered with Bristol Myers Squibb PD-L1/VEGF-A Complex Formation and Internalisation VEGF-A Neutralisation VEGF-A Neutralisation Tumor cellT cell PD-L1 Blockade PD-1 Anti-VEGF-A LALA silencing mutations Anti-PD-L1 VHH Stable linkers Differentiated MoA Enhanced dual blockade of PD-L1 and VEGF-A, mediated by a single bispecific molecule Clinical evidence Studied in 18 tumor types and 1600+ patients Impressive efficacy signals across tumor types Clinical benefit observed in several tumor types, e.g. TNBC, NSCLC, regardless of PD-L1 expression in global population Global data consistent with data generated in China Development status 8 pivotal studies ongoing by year-end 2026 12+ combination trials with chemotherapy 10+ novel-novel combinations trials
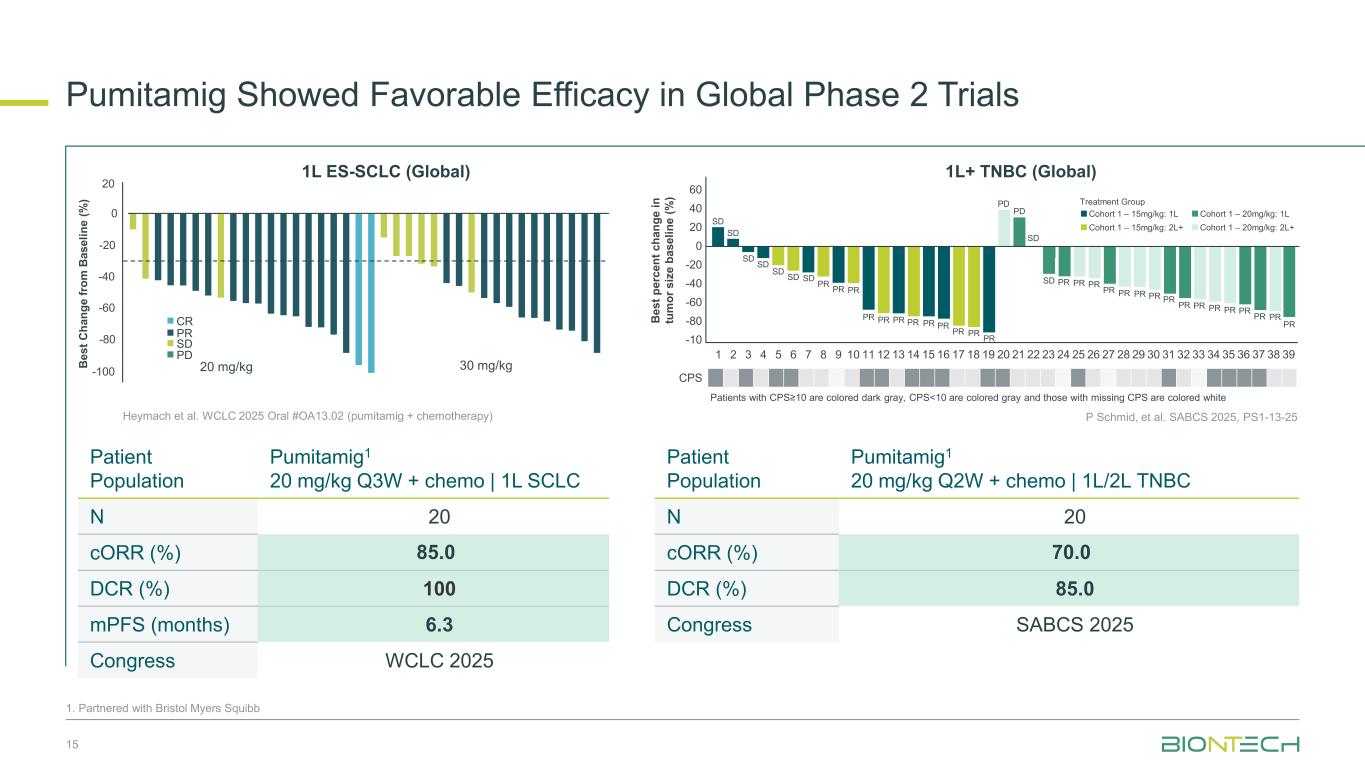
Pumitamig Showed Favorable Efficacy in Global Phase 2 Trials 15 1. Partnered with Bristol Myers Squibb Patient Population Pumitamig1 20 mg/kg Q3W + chemo | 1L SCLC N 20 cORR (%) 85.0 DCR (%) 100 mPFS (months) 6.3 Congress WCLC 2025 Patient Population Pumitamig1 20 mg/kg Q2W + chemo | 1L/2L TNBC N 20 cORR (%) 70.0 DCR (%) 85.0 Congress SABCS 2025 20 0 -20 -40 -60 -80 -100 30 mg/kg20 mg/kgB es t C ha ng e fr om B as el in e (% ) PD SD PR CR Heymach et al. WCLC 2025 Oral #OA13.02 (pumitamig + chemotherapy) 1L ES-SCLC (Global) P Schmid, et al. SABCS 2025, PS1-13-25 B es t p er ce nt c ha ng e in tu m or s iz e ba se lin e (% ) 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 60 40 20 0 -20 -40 -60 -80 -10 SD SD SD SD SD SD SD PR PR PR PR PR PR PR PR PR PR PR PR PD PD SD SD PR PR PR PR PR PR PR PR PR PR PR PR PR PR PR PR Treatment Group Cohort 1 – 15mg/kg: 1L Cohort 1 – 15mg/kg: 2L+ Cohort 1 – 20mg/kg: 1L Cohort 1 – 20mg/kg: 2L+ CPS Patients with CPS≥10 are colored dark gray, CPS<10 are colored gray and those with missing CPS are colored white 1L+ TNBC (Global)

SCLC 1L Ph3 (Global) 2L Ph3 (China) 1L/2L Ph2 (Global) NSCLC 1L Ph2/3 (Global) 2L Ph2 (Global) 2L EGFRmut Ph2 (China) IIT neoadjuvant (China) TNBC 1L Ph3 trial (Global) 1L Ph3 (China) ElevateEstablish Expand Executing a Parallel Three-Wave Strategy to Build a Proprietary IO Franchise 16 1. Partnered with Bristol Myers Squibb. Combining with ADCs targeting: HER2 TROP2 B7H3 Exploring potential synergies with our IO agents EpCAM/4-1BB TIGIT/PVRIG mRNA cancer immunotherapy Registrational-Intent 1L Gastric Ph2/3 (Global) 1L CRC Ph2/3 (Global) 1L HNSCC Phase 2/3 (Global) Signal-Seeking 1L PDAC Ph2 (China) 1L GBM Ph2 (China) 1L RCC Phase 1/2 (Global) 1L CRC Ph2 (China) 1L HCC Phase 1/2 (Global) 1L HCC Ph2 (China) 1L MPM Ph2 (China) 1L NEN Ph2 (China) HNSCC, RCC, CC, PROC, EC, Melanoma Ph1/2 (China) Novel targets Foundational Registrations Registrational Trials with Pumitamig1 Ongoing in 3 High-Impact Tumors Broad Pan-Tumor Applicability With Standard-of-Care Chemotherapy 12+ Studies Exploring Pumitamig1 in 10+ New Indications Potential New Standards of Care 10+ Novel-Novel Combinations EGFR/HER3 HER3
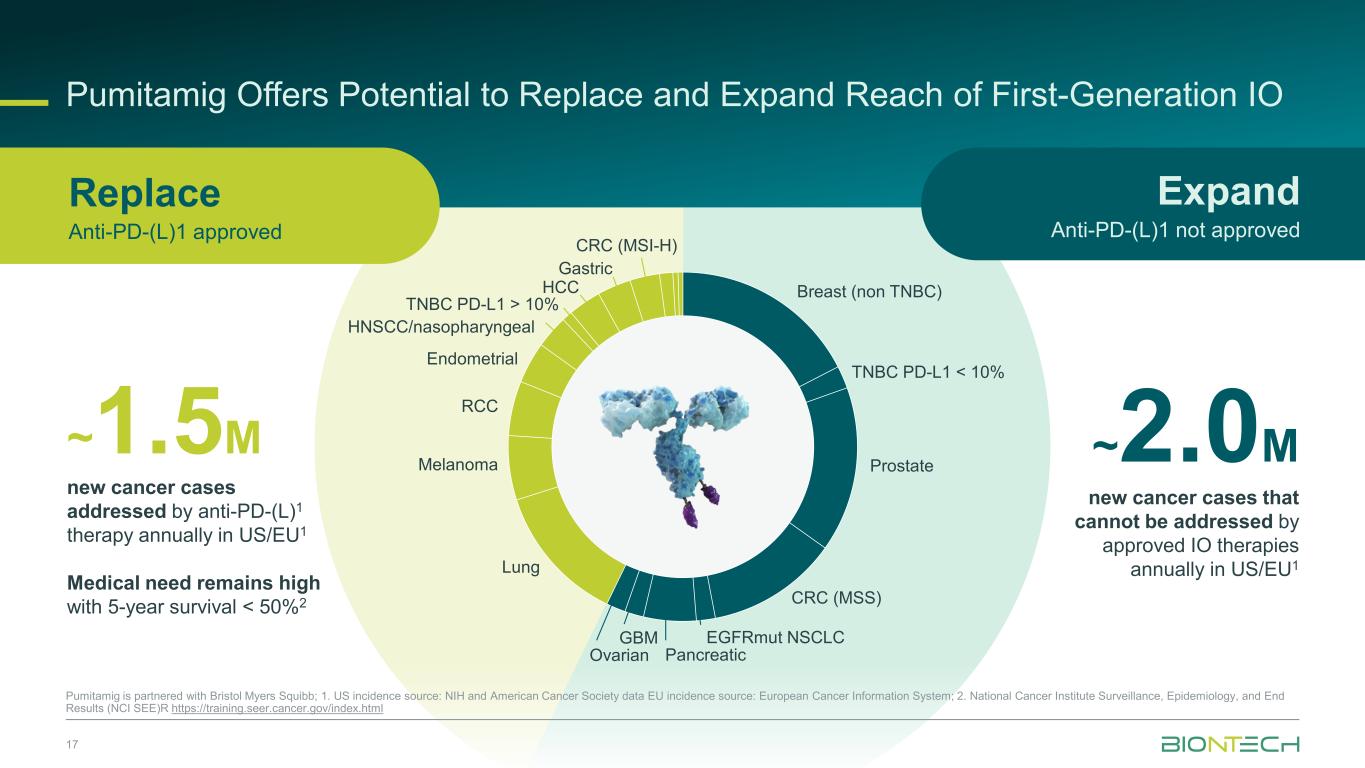
Breast (non TNBC) TNBC PD-L1 < 10% Prostate CRC (MSS) EGFRmut NSCLC Pancreatic GBM Ovarian Lung Melanoma RCC Endometrial HNSCC/nasopharyngeal TNBC PD-L1 > 10% HCC Gastric CRC (MSI-H) Pumitamig Offers Potential to Replace and Expand Reach of First-Generation IO 17 Pumitamig is partnered with Bristol Myers Squibb; 1. US incidence source: NIH and American Cancer Society data EU incidence source: European Cancer Information System; 2. National Cancer Institute Surveillance, Epidemiology, and End Results (NCI SEE)R https://training.seer.cancer.gov/index.html ~1.5M new cancer cases addressed by anti-PD-(L)1 therapy annually in US/EU1 Medical need remains high with 5-year survival < 50%2 ~2.0M new cancer cases that cannot be addressed by approved IO therapies annually in US/EU1 Expand Anti-PD-(L)1 not approved Replace Anti-PD-(L)1 approved
Unique Opportunity to Shape Next Wave of Solid Tumor Therapy 18 Partnered with: 1. Bristol Myers Squibb; 2. OncoC4; 3. Duality Bio Gotistobart2 TME-selective regulatory T cell-depleting antibody targeting CTLA-4
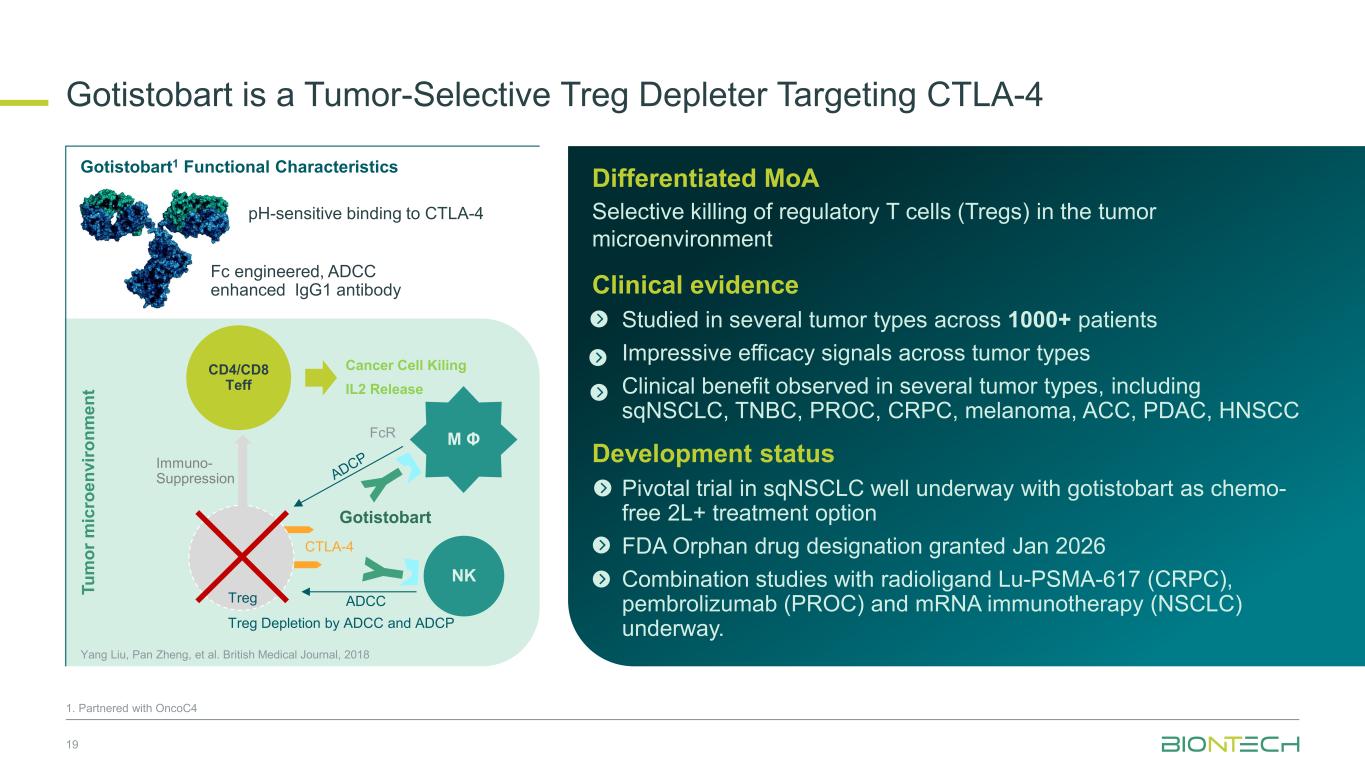
Gotistobart is a Tumor-Selective Treg Depleter Targeting CTLA-4 19 1. Partnered with OncoC4 Tu m or m ic ro en vi ro nm en t NK M Φ CTLA-4 Gotistobart FcR ADCCTreg CD4/CD8 Teff Yang Liu, Pan Zheng, et al. British Medical Journal, 2018 Immuno- Suppression Cancer Cell Kiling IL2 Release Differentiated MoA Selective killing of regulatory T cells (Tregs) in the tumor microenvironment Clinical evidence Studied in several tumor types across 1000+ patients Impressive efficacy signals across tumor types Clinical benefit observed in several tumor types, including sqNSCLC, TNBC, PROC, CRPC, melanoma, ACC, PDAC, HNSCC Development status Pivotal trial in sqNSCLC well underway with gotistobart as chemo- free 2L+ treatment option FDA Orphan drug designation granted Jan 2026 Combination studies with radioligand Lu-PSMA-617 (CRPC), pembrolizumab (PROC) and mRNA immunotherapy (NSCLC) underway.Treg Depletion by ADCC and ADCP Gotistobart1 Functional Characteristics Fc engineered, ADCC enhanced IgG1 antibody pH-sensitive binding to CTLA-4
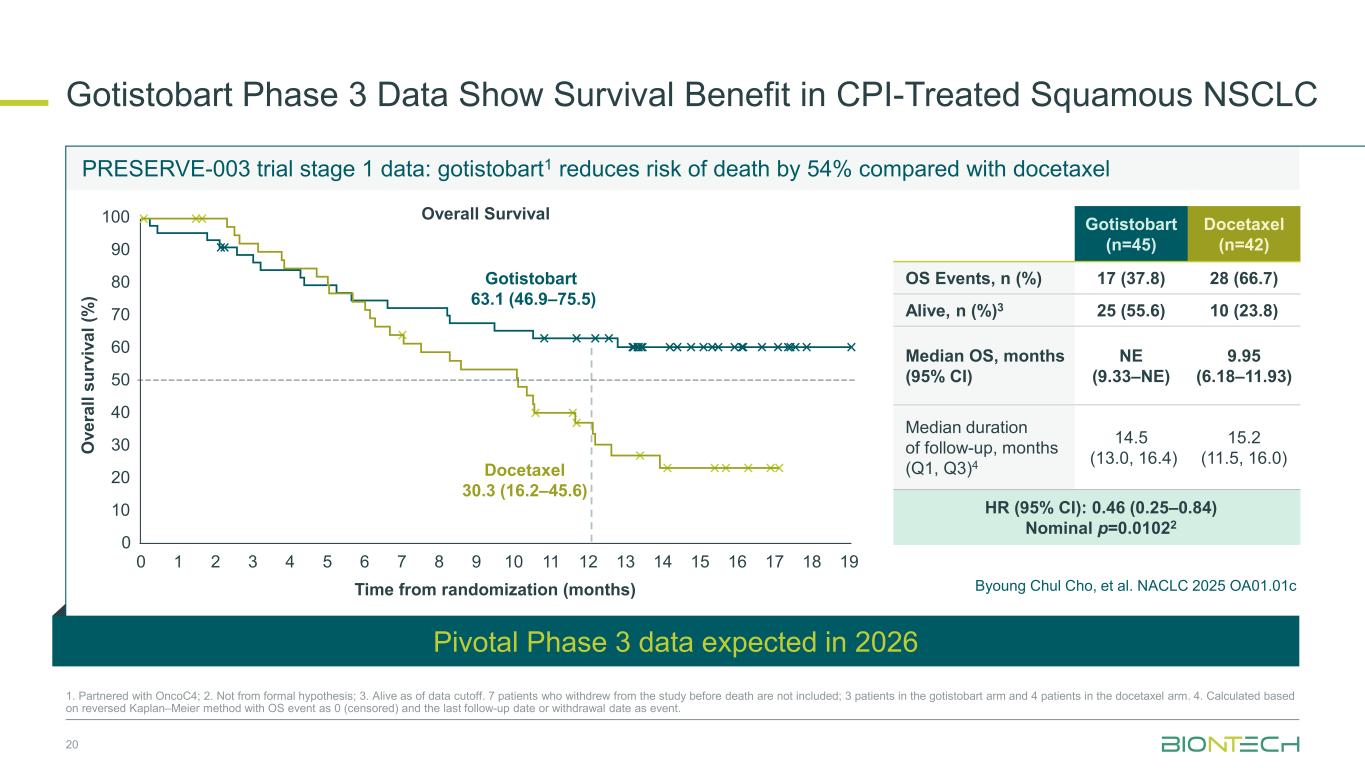
Docetaxel 30.3 (16.2–45.6) O ve ra ll su rv iv al (% ) 0 10 20 30 40 50 60 70 80 90 100 Gotistobart 63.1 (46.9–75.5) Overall Survival PRESERVE-003 trial stage 1 data: gotistobart1 reduces risk of death by 54% compared with docetaxel 20 1. Partnered with OncoC4; 2. Not from formal hypothesis; 3. Alive as of data cutoff. 7 patients who withdrew from the study before death are not included; 3 patients in the gotistobart arm and 4 patients in the docetaxel arm. 4. Calculated based on reversed Kaplan–Meier method with OS event as 0 (censored) and the last follow-up date or withdrawal date as event. 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 Time from randomization (months) Pivotal Phase 3 data expected in 2026 Gotistobart Phase 3 Data Show Survival Benefit in CPI-Treated Squamous NSCLC Gotistobart (n=45) Docetaxel (n=42) OS Events, n (%) 17 (37.8) 28 (66.7) Alive, n (%)3 25 (55.6) 10 (23.8) Median OS, months (95% CI) NE (9.33–NE) 9.95 (6.18–11.93) Median duration of follow-up, months (Q1, Q3)4 14.5 (13.0, 16.4) 15.2 (11.5, 16.0) HR (95% CI): 0.46 (0.25–0.84) Nominal p=0.01022 Byoung Chul Cho, et al. NACLC 2025 OA01.01c

Squamous NSCLC Remains an Area of High Unmet Need 21 1. By 2030 in US & EU5, CancerMPact; 2. Clarivate / Clarivate Survey; 3. Partnered with OncoC4 Limited treatment options for squamous NSCLC patients without actionable genetic alterations Metastatic squamous NSCLC seen as #1 area of unmet need in NSCLC2 In 2L, current chemo-based SoC offers only 10 months median OS in clinical trials <25% patients respond to 2L chemo-based SOC (docetaxel ± ramucirumab) Multiple Ph3 trials failed to improve therapeutic outcome in 2L squamous NSCLC in recent years ~55k squamous NSCLC patients initiate 1L treatment (non-AGA population)1 ~30% continue into 2L treatment and could be eligible for gotistobart3
Unique Opportunity to Shape Next Wave of Solid Tumor Therapy 22 Partnered with: 1. Bristol Myers Squibb; 2. OncoC4; 3. Duality Bio BNT324/DB-13113 Novel pan-tumor ADC targeting B7H3 with favorable safety profile

BNT324/DB-1311 B7H3-Targeted ADC with Pan-Tumor Potential 23 1. Partnered with DualityBio; 2. CC and PROC: Chang et al ESMO Asia 2025; CRPC: Parsonson et al ASCO 2025; SCLC, NSCLC, ESCC, melanoma, HCC, HNSCC: ESMO Asia 2024 3. Human Protein Atlas B7H3 is overexpressed in multiple tumor types B7H3-targeting ADC with DAR=6 and novel topoisomerase inhibitor Clinical evidence 600+ patients treated with BNT324/DB-13111 across 10+ indications Antitumor activity in multiple tumor types2 Favorable safety profile Development status Phase 3 in 1L mCRPC active Monotherapy and pumitamig combination trials ongoing B7H3 is overexpressed in multiple tumor types Prostate NSCLC AGA- NSCLC EGFRm SCLC HR+ BC TNBC CRC Gastric Cervical Ovarian PDAC HNSCC HCC Melanoma B7H3 Expression Level3 Development Status Ph3 Ph1/2 Ph1/2 Ph1/2 Ph1/2 Ph1/2 Ph1/2 Ph1/2 Ph1/2 Ph1/2 Expression level High Medium/Low Very low/None
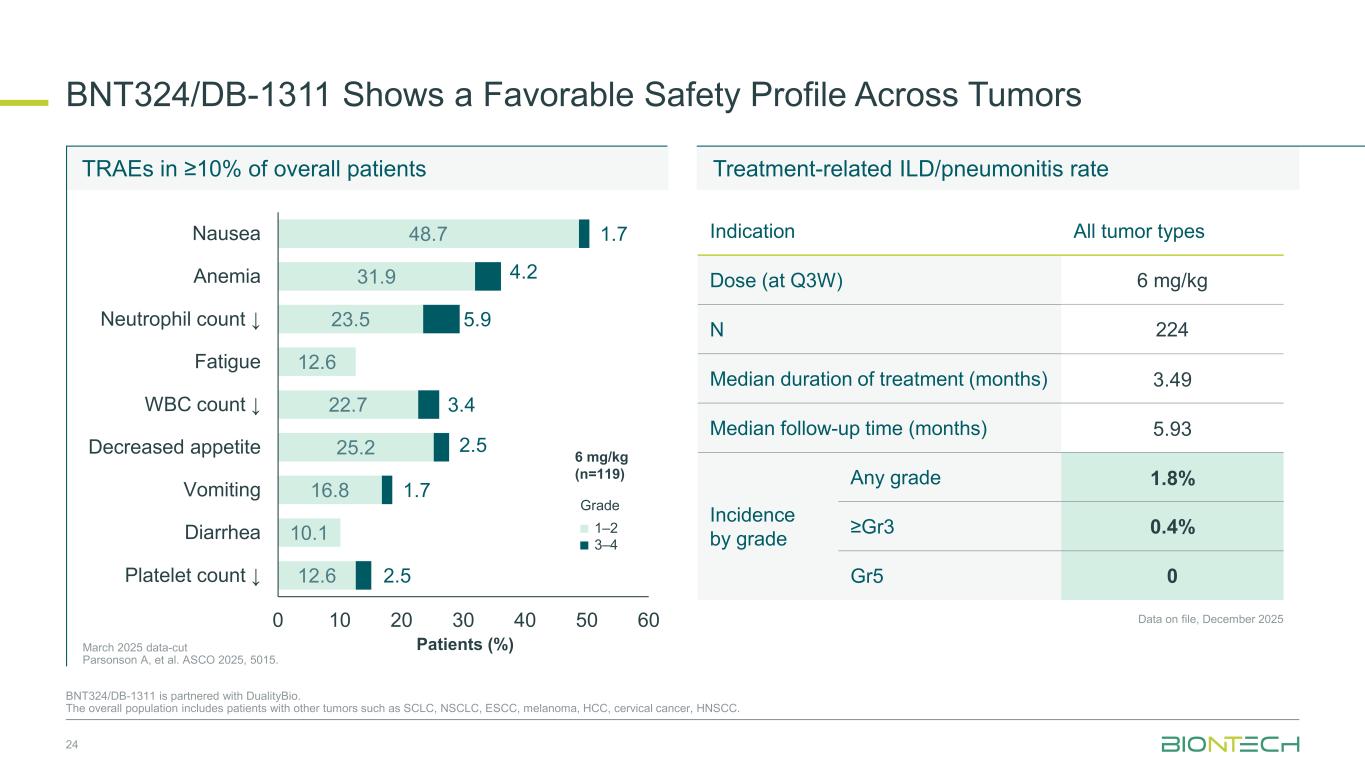
BNT324/DB-1311 Shows a Favorable Safety Profile Across Tumors 24 BNT324/DB-1311 is partnered with DualityBio. The overall population includes patients with other tumors such as SCLC, NSCLC, ESCC, melanoma, HCC, cervical cancer, HNSCC. 48.7 31.9 23.5 12.6 22.7 25.2 16.8 10.1 12.6 1.7 4.2 5.9 3.4 2.5 1.7 2.5 0 10 20 30 40 50 60 Nausea Anemia Neutrophil count ↓ Fatigue WBC count ↓ Decreased appetite Vomiting Diarrhea Platelet count ↓ Patients (%) 1–2 3–4 Grade 6 mg/kg (n=119) Treatment-related ILD/pneumonitis rate Data on file, December 2025 March 2025 data-cut Parsonson A, et al. ASCO 2025, 5015. Indication All tumor types Dose (at Q3W) 6 mg/kg N 224 Median duration of treatment (months) 3.49 Median follow-up time (months) 5.93 Incidence by grade Any grade 1.8% ≥Gr3 0.4% Gr5 0 TRAEs in ≥10% of overall patients
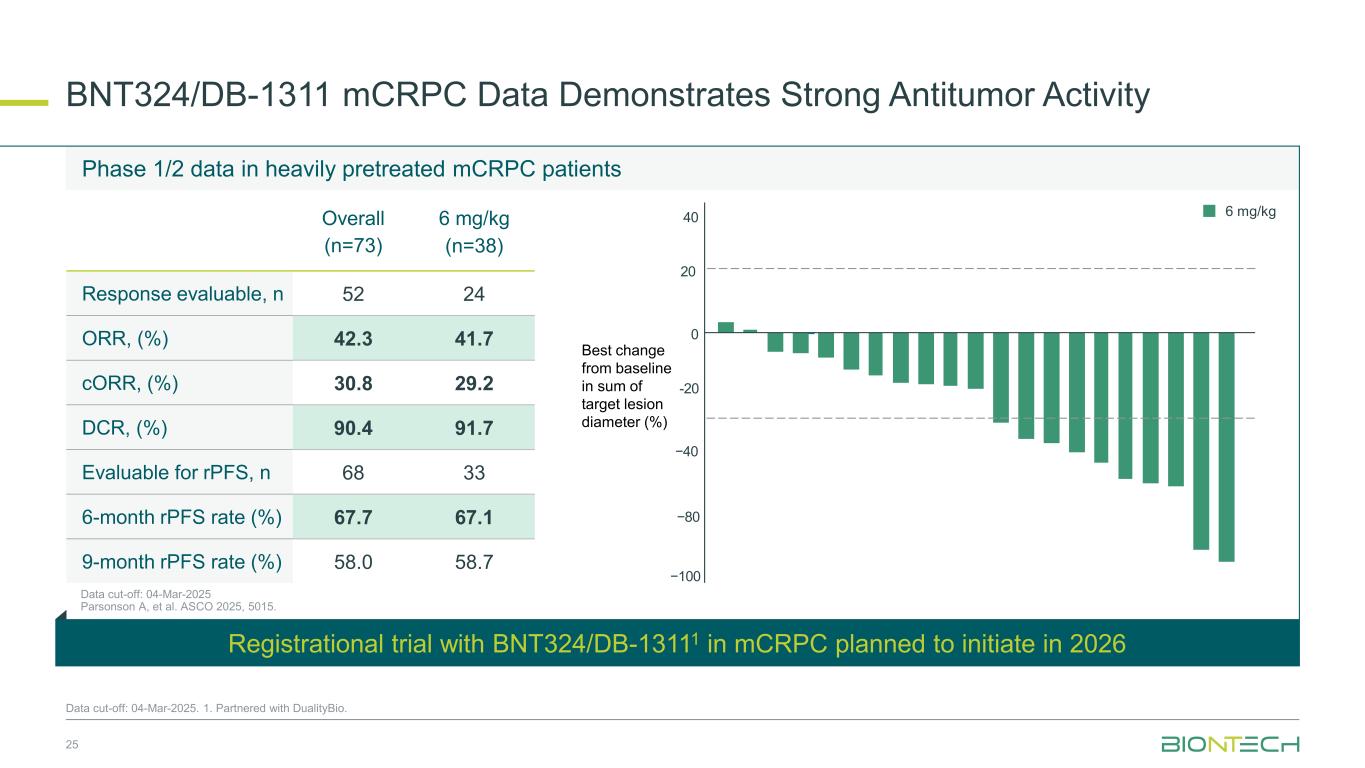
BNT324/DB-1311 mCRPC Data Demonstrates Strong Antitumor Activity 25 Data cut-off: 04-Mar-2025. 1. Partnered with DualityBio. Phase 1/2 data in heavily pretreated mCRPC patients Best change from baseline in sum of target lesion diameter (%) −80 −40 0 40 20 -20 −100 Registrational trial with BNT324/DB-13111 in mCRPC planned to initiate in 2026 Overall (n=73) 6 mg/kg (n=38) Response evaluable, n 52 24 ORR, (%) 42.3 41.7 cORR, (%) 30.8 29.2 DCR, (%) 90.4 91.7 Evaluable for rPFS, n 68 33 6-month rPFS rate (%) 67.7 67.1 9-month rPFS rate (%) 58.0 58.7 6 mg/kg Data cut-off: 04-Mar-2025 Parsonson A, et al. ASCO 2025, 5015.

Evolving mCRPC Landscape Offers Significant Opportunity For New Treatments 26 Source: Evaluate Pharma, DRG, CancerMpact Many patients are ineligible for docetaxel or wish to delay or avoid chemo mCRPC is a leading cause of cancer related mortality Need remains for easily accessible treatment options in early setting, that are safe and provide more durable responses Docetaxel becoming 1L therapy of choice 80k drug treated mCRPC patients in US by 2040 ~$22B Expected global prostate cancer market by 2030
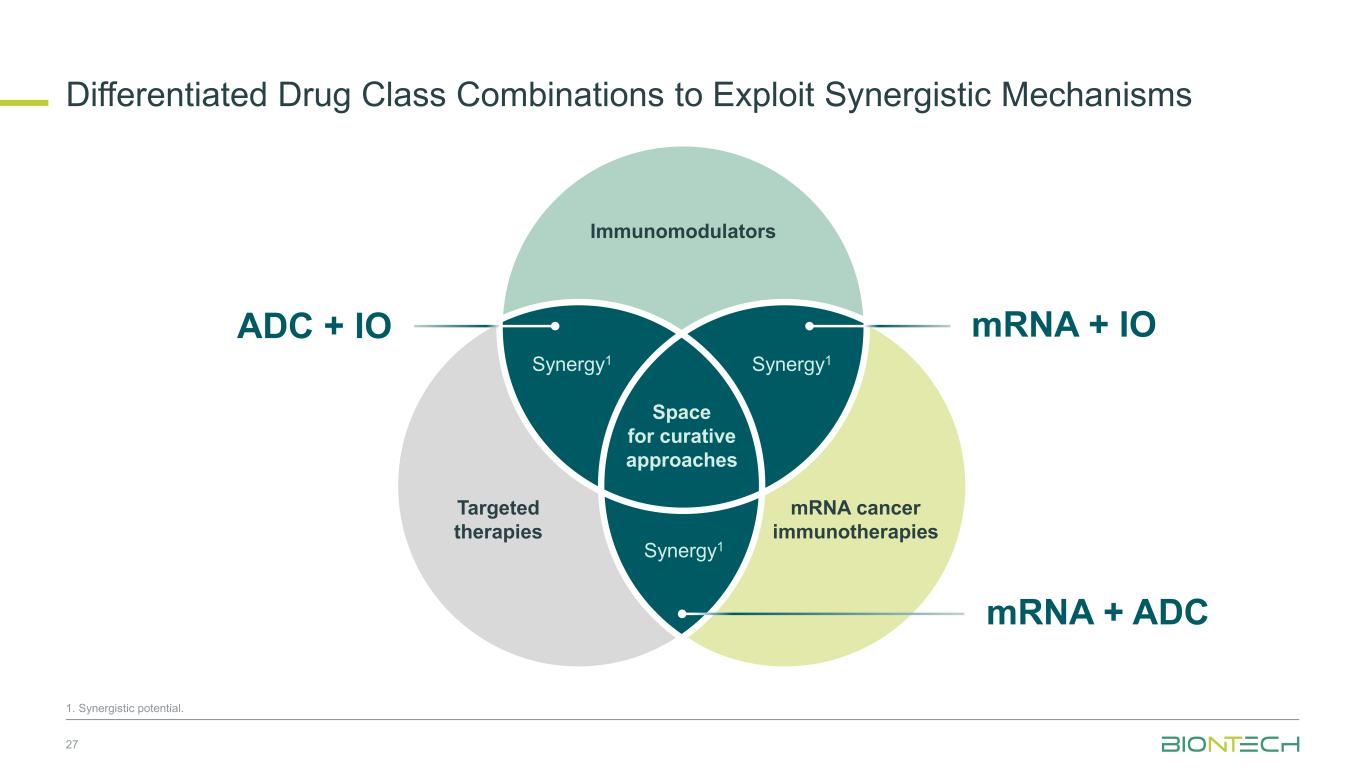
Differentiated Drug Class Combinations to Exploit Synergistic Mechanisms 27 1. Synergistic potential. Space for curative approaches Immunomodulators Targeted therapies Synergy1Synergy1 Synergy1 mRNA cancer immunotherapies mRNA + IO mRNA + ADC ADC + IO
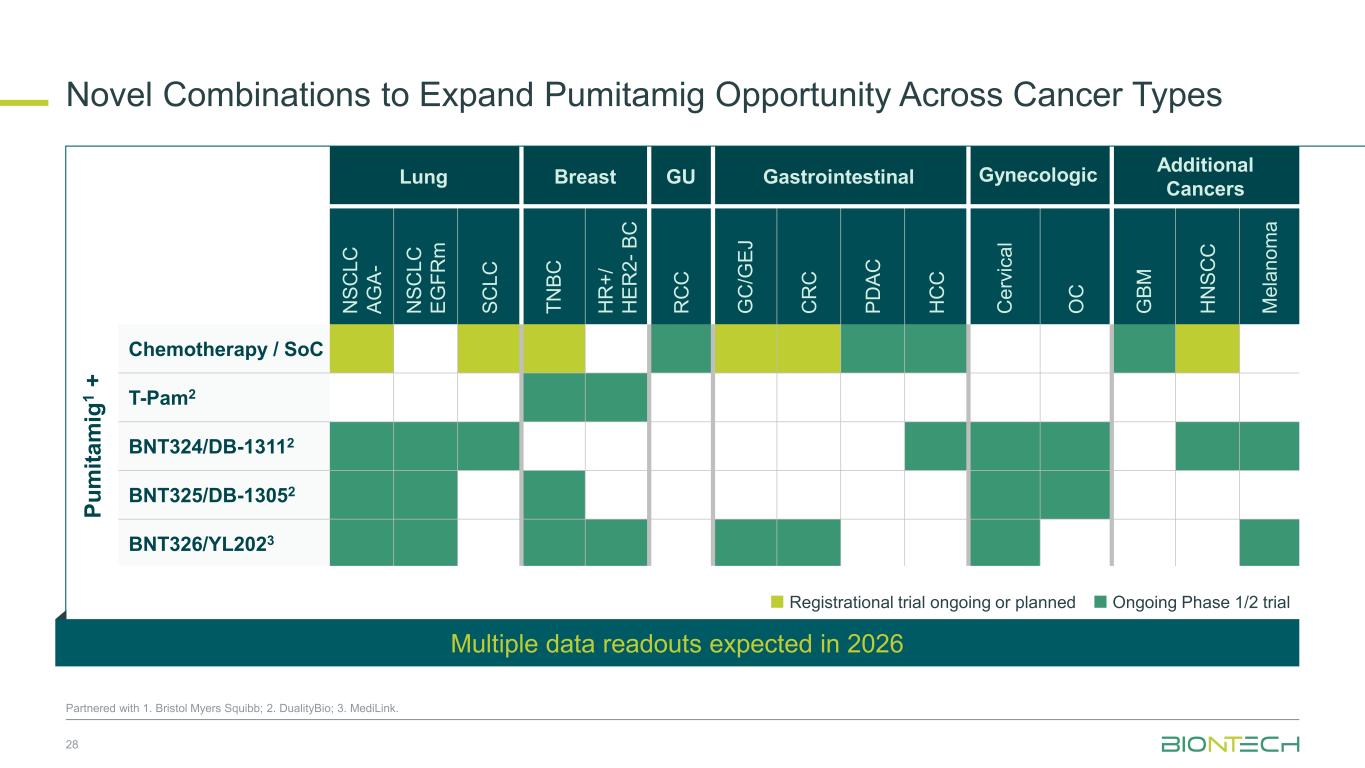
Lung Breast GU Gastrointestinal Gynecologic Additional Cancers N SC LC AG A- N SC LC EG FR m SC LC TN BC H R +/ H ER 2- B C R C C G C /G EJ C R C PD AC H C C C er vi ca l O C G BM H N SC C M el an om a Pu m ita m ig 1 + Chemotherapy / SoC T-Pam2 BNT324/DB-13112 BNT325/DB-13052 BNT326/YL2023 Novel Combinations to Expand Pumitamig Opportunity Across Cancer Types 28 Partnered with 1. Bristol Myers Squibb; 2. DualityBio; 3. MediLink. Registrational trial ongoing or planned Ongoing Phase 1/2 trial Multiple data readouts expected in 2026
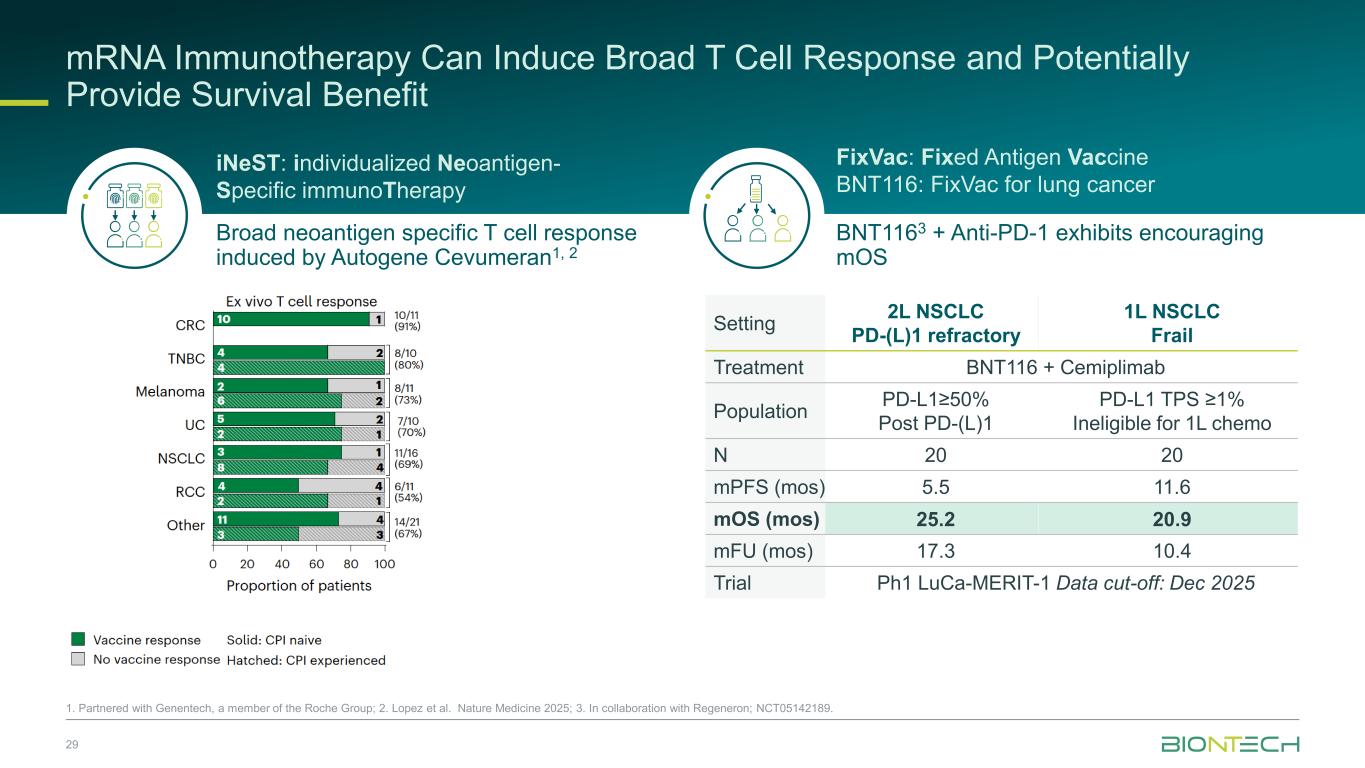
mRNA Immunotherapy Can Induce Broad T Cell Response and Potentially Provide Survival Benefit 29 1. Partnered with Genentech, a member of the Roche Group; 2. Lopez et al. Nature Medicine 2025; 3. In collaboration with Regeneron; NCT05142189. Setting 2L NSCLC PD-(L)1 refractory 1L NSCLC Frail Treatment BNT116 + Cemiplimab Population PD-L1≥50% Post PD-(L)1 PD-L1 TPS ≥1% Ineligible for 1L chemo N 20 20 mPFS (mos) 5.5 11.6 mOS (mos) 25.2 20.9 mFU (mos) 17.3 10.4 Trial Ph1 LuCa-MERIT-1 Data cut-off: Dec 2025 Broad neoantigen specific T cell response induced by Autogene Cevumeran1, 2 BNT1163 + Anti-PD-1 exhibits encouraging mOS iNeST: individualized Neoantigen- Specific immunoTherapy FixVac: Fixed Antigen Vaccine BNT116: FixVac for lung cancer
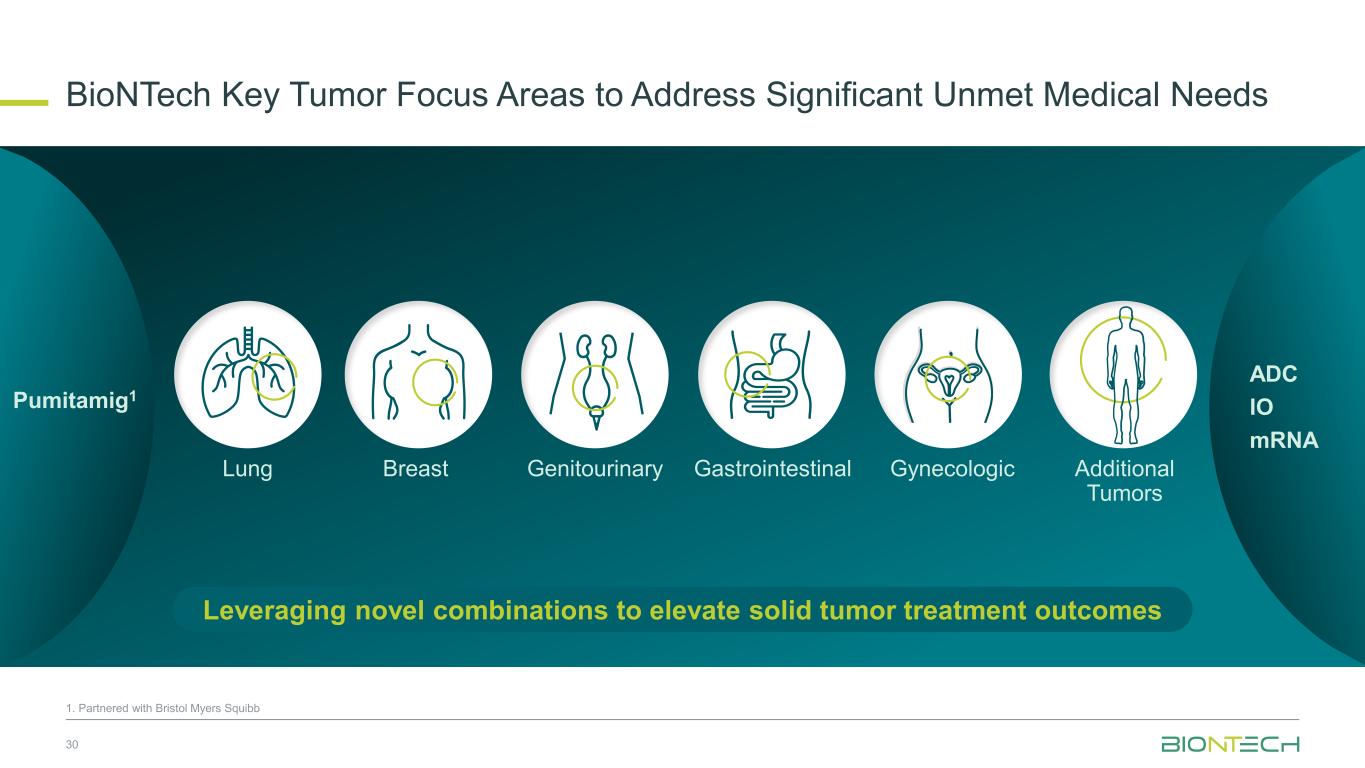
BioNTech Key Tumor Focus Areas to Address Significant Unmet Medical Needs 30 Leveraging novel combinations to elevate solid tumor treatment outcomes Lung Breast Genitourinary Gastrointestinal Gynecologic Additional Tumors Pumitamig1 ADC IO mRNA 1. Partnered with Bristol Myers Squibb
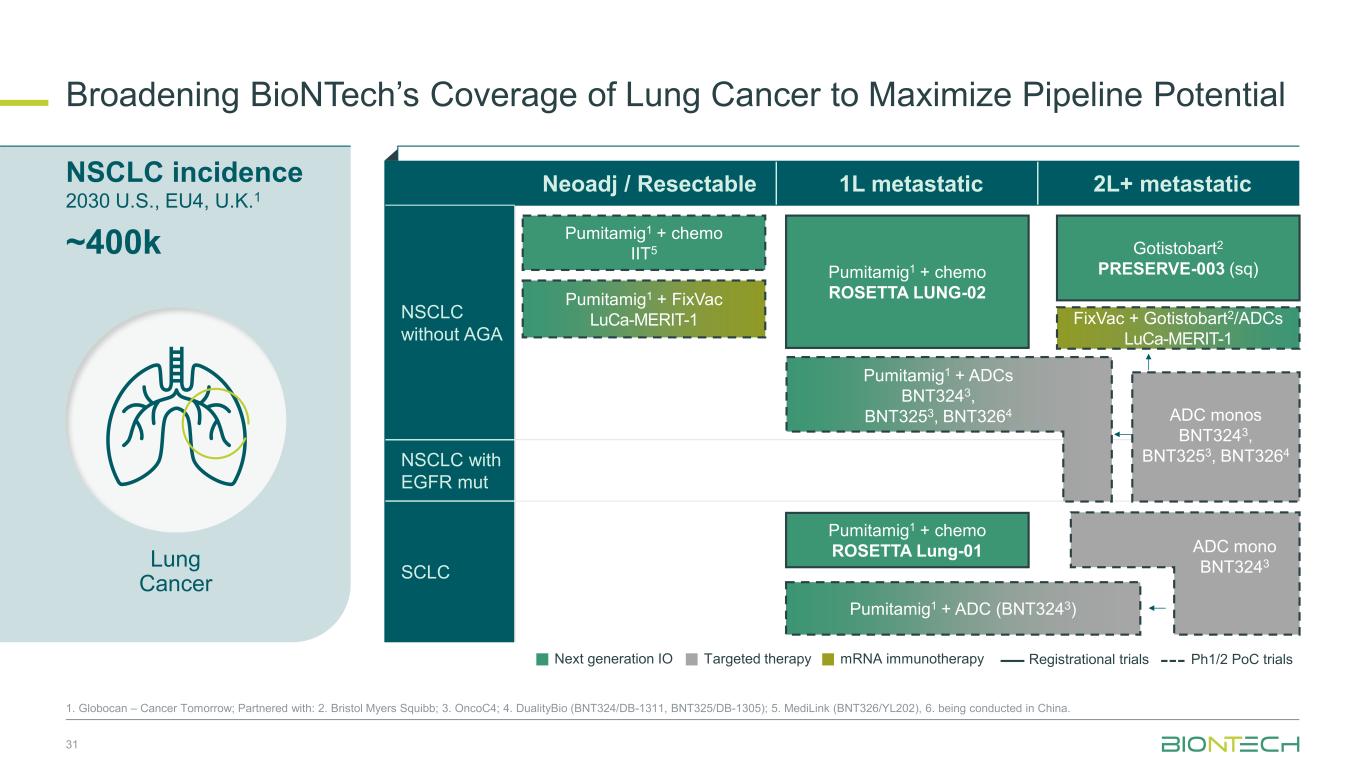
NSCLC incidence 2030 U.S., EU4, U.K.1 Broadening BioNTech’s Coverage of Lung Cancer to Maximize Pipeline Potential 31 1. Globocan – Cancer Tomorrow; Partnered with: 2. Bristol Myers Squibb; 3. OncoC4; 4. DualityBio (BNT324/DB-1311, BNT325/DB-1305); 5. MediLink (BNT326/YL202), 6. being conducted in China. ~400k Neoadj / Resectable 1L metastatic 2L+ metastatic NSCLC without AGA NSCLC with EGFR mut SCLC Gotistobart2 PRESERVE-003 (sq) FixVac + Gotistobart2/ADCs LuCa-MERIT-1 Pumitamig1 + chemo ROSETTA Lung-01 Pumitamig1 + chemo IIT5 Pumitamig1 + chemo ROSETTA LUNG-02 ADC monos BNT3243, BNT3253, BNT3264 Pumitamig1 + FixVac LuCa-MERIT-1 Pumitamig1 + ADCs BNT3243, BNT3253, BNT3264 ADC mono BNT3243 Pumitamig1 + ADC (BNT3243) Lung Cancer Next generation IO Targeted therapy mRNA immunotherapy Registrational trials Ph1/2 PoC trials
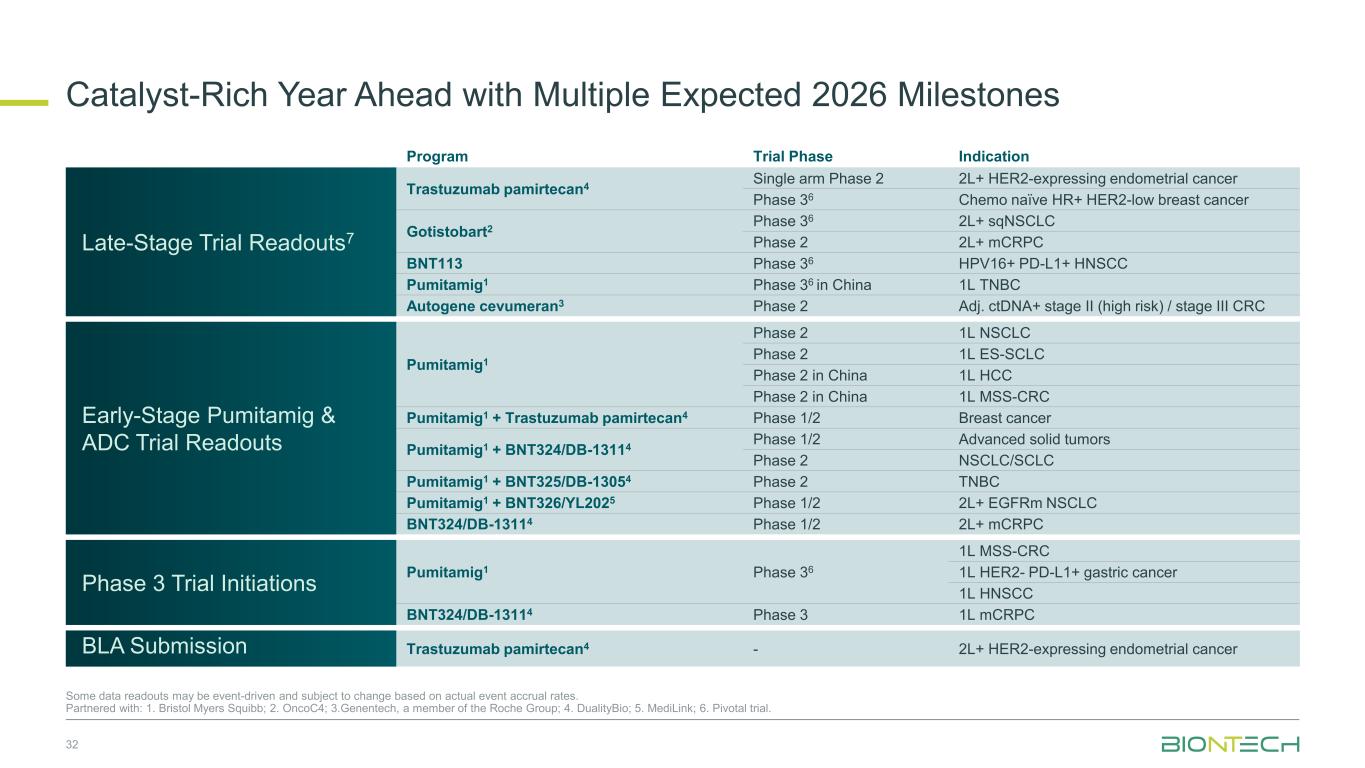
Catalyst-Rich Year Ahead with Multiple Expected 2026 Milestones 32 Some data readouts may be event-driven and subject to change based on actual event accrual rates. Partnered with: 1. Bristol Myers Squibb; 2. OncoC4; 3.Genentech, a member of the Roche Group; 4. DualityBio; 5. MediLink; 6. Pivotal trial. Program Trial Phase Indication Late-Stage Trial Readouts7 Trastuzumab pamirtecan4 Single arm Phase 2 2L+ HER2-expressing endometrial cancer Phase 36 Chemo naïve HR+ HER2-low breast cancer Gotistobart2 Phase 36 2L+ sqNSCLC Phase 2 2L+ mCRPC BNT113 Phase 36 HPV16+ PD-L1+ HNSCC Pumitamig1 Phase 36 in China 1L TNBC Autogene cevumeran3 Phase 2 Adj. ctDNA+ stage II (high risk) / stage III CRC Early-Stage Pumitamig & ADC Trial Readouts Pumitamig1 Phase 2 1L NSCLC Phase 2 1L ES-SCLC Phase 2 in China 1L HCC Phase 2 in China 1L MSS-CRC Pumitamig1 + Trastuzumab pamirtecan4 Phase 1/2 Breast cancer Pumitamig1 + BNT324/DB-13114 Phase 1/2 Advanced solid tumors Phase 2 NSCLC/SCLC Pumitamig1 + BNT325/DB-13054 Phase 2 TNBC Pumitamig1 + BNT326/YL2025 Phase 1/2 2L+ EGFRm NSCLC BNT324/DB-13114 Phase 1/2 2L+ mCRPC Phase 3 Trial Initiations Pumitamig1 Phase 36 1L MSS-CRC 1L HER2- PD-L1+ gastric cancer 1L HNSCC BNT324/DB-13114 Phase 3 1L mCRPC BLA Submission Trastuzumab pamirtecan4 - 2L+ HER2-expressing endometrial cancer
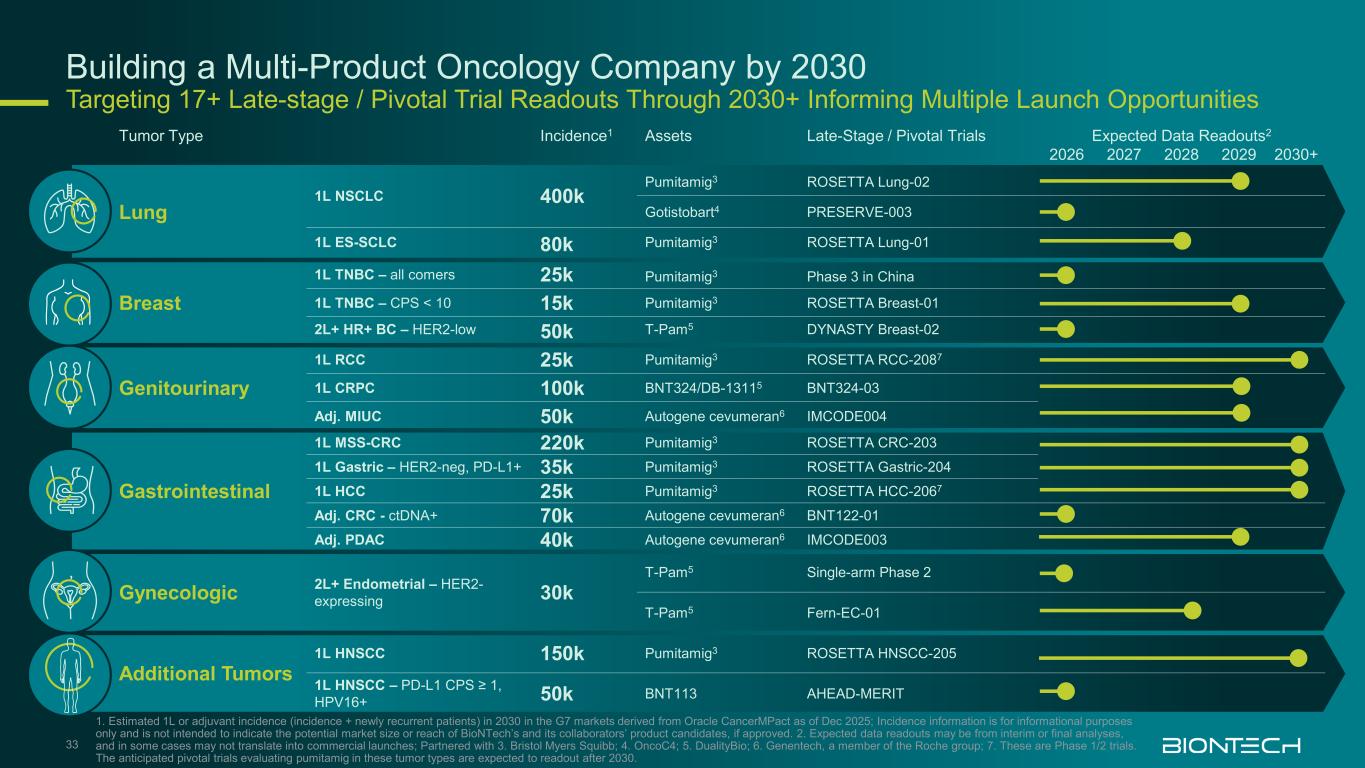
Building a Multi-Product Oncology Company by 2030 Targeting 17+ Late-stage / Pivotal Trial Readouts Through 2030+ Informing Multiple Launch Opportunities 33 1. Estimated 1L or adjuvant incidence (incidence + newly recurrent patients) in 2030 in the G7 markets derived from Oracle CancerMPact as of Dec 2025; Incidence information is for informational purposes only and is not intended to indicate the potential market size or reach of BioNTech’s and its collaborators’ product candidates, if approved. 2. Expected data readouts may be from interim or final analyses, and in some cases may not translate into commercial launches; Partnered with 3. Bristol Myers Squibb; 4. OncoC4; 5. DualityBio; 6. Genentech, a member of the Roche group; 7. These are Phase 1/2 trials. The anticipated pivotal trials evaluating pumitamig in these tumor types are expected to readout after 2030. Tumor Type Incidence1 Assets Late-Stage / Pivotal Trials Expected Data Readouts2 2026 2027 2028 2029 2030+ Lung 1L NSCLC 400k Pumitamig3 ROSETTA Lung-02 Gotistobart4 PRESERVE-003 1L ES-SCLC 80k Pumitamig3 ROSETTA Lung-01 Breast 1L TNBC – all comers 25k Pumitamig3 Phase 3 in China 1L TNBC – CPS < 10 15k Pumitamig3 ROSETTA Breast-01 2L+ HR+ BC – HER2-low 50k T-Pam5 DYNASTY Breast-02 Genitourinary 1L RCC 25k Pumitamig3 ROSETTA RCC-2087 1L CRPC 100k BNT324/DB-13115 BNT324-03 Adj. MIUC 50k Autogene cevumeran6 IMCODE004 Gastrointestinal 1L MSS-CRC 220k Pumitamig3 ROSETTA CRC-203 1L Gastric – HER2-neg, PD-L1+ 35k Pumitamig3 ROSETTA Gastric-204 1L HCC 25k Pumitamig3 ROSETTA HCC-2067 Adj. CRC - ctDNA+ 70k Autogene cevumeran6 BNT122-01 Adj. PDAC 40k Autogene cevumeran6 IMCODE003 Gynecologic 2L+ Endometrial – HER2- expressing 30k T-Pam5 Single-arm Phase 2 T-Pam5 Fern-EC-01 Additional Tumors 1L HNSCC 150k Pumitamig3 ROSETTA HNSCC-205 1L HNSCC – PD-L1 CPS ≥ 1, HPV16+ 50k BNT113 AHEAD-MERIT
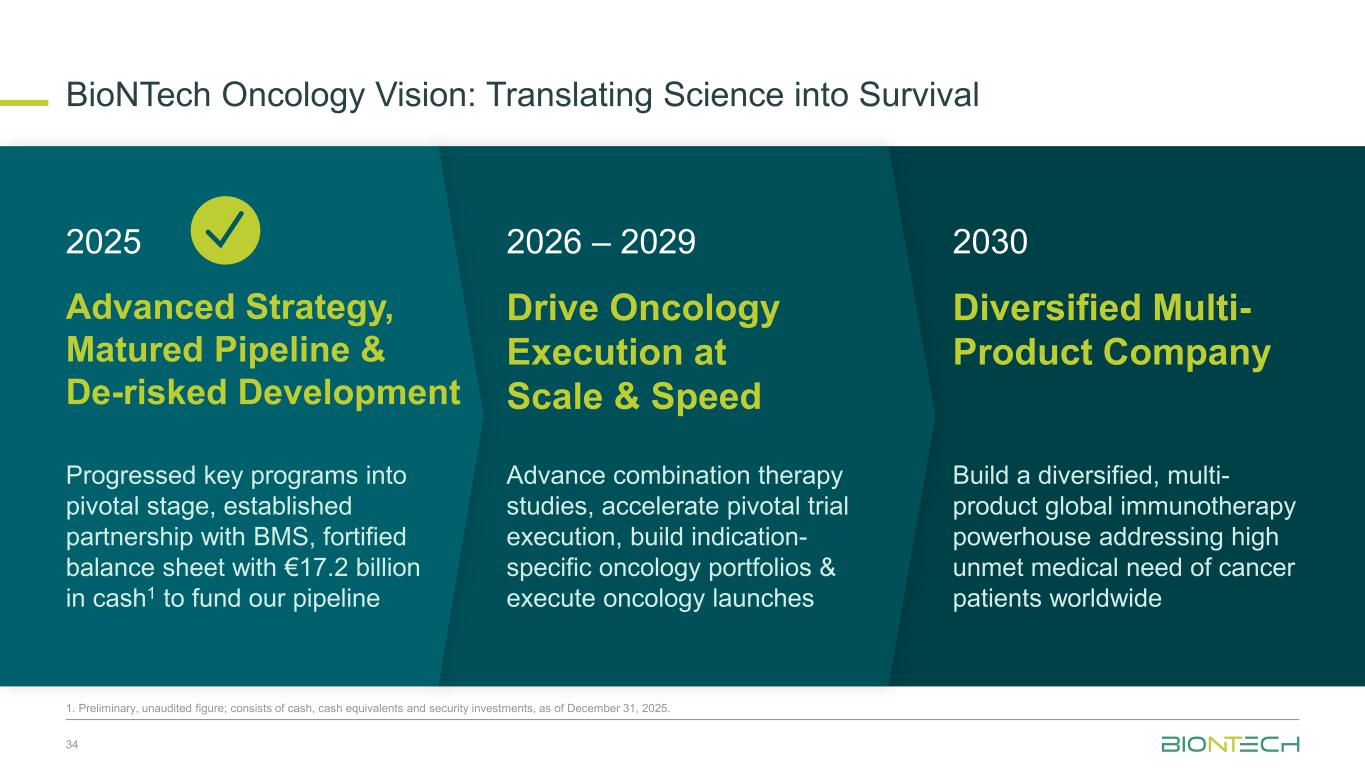
2025 2026 – 2029 2030 Advanced Strategy, Matured Pipeline & De-risked Development Drive Oncology Execution at Scale & Speed Diversified Multi- Product Company Progressed key programs into pivotal stage, established partnership with BMS, fortified balance sheet with €17.2 billion in cash1 to fund our pipeline Advance combination therapy studies, accelerate pivotal trial execution, build indication- specific oncology portfolios & execute oncology launches Build a diversified, multi- product global immunotherapy powerhouse addressing high unmet medical need of cancer patients worldwide BioNTech Oncology Vision: Translating Science into Survival 34 1. Preliminary, unaudited figure; consists of cash, cash equivalents and security investments, as of December 31, 2025.

Thank you
Abbreviation Directory 36 4-1BB CD137 FixVac Fixed Antigen Vaccine (sq) NSCLC (sqamous) Non-small cell lung cancer n L nth line (m)FU (median) Follow-up time OC Ovarian cancer a Anti G7 Canada, France, Germany, Italy, Japan, Great Britain, USA (c)ORR (confirmed) Objective response rate ACC Adenoid cystic carcinoma GBM Glioblastoma OS Overall survival (bs)ADC (bispecific) Antibody-drug conjugate GC/GEJ Gastric/Gastro-esophageal junction cancer P&L Profit and loss statement ADCC Antibody-dependent cell-mediated cytotoxicity GU Genitourinary PD Progressive disease ADCP Antibody-dependent cellular phagocytosis HCC Hepatocellular carcinoma PD-(L)1 Programmed cell death protein (ligand) 1 Adj. Adjuvant HER2 (or 3) Human epidermal growth factor receptor 2 (or 3) PDAC Pancreatic ductal adenocarcinoma AGA Actionable oncogenic alteration HNSCC Head and neck squamous cell carcinoma (m)PFS (median) Progression-free survival AI Artificial intelligence HPV16 Human papilloma virus 16 PoC Proof of concept ASCO American Society of Clinical Oncology HR Hazard ratio / Hormone receptor PR Partial response B7-H3 B7 Homolog 3 IgG1 Immunoglobulin G1 PROC Platinum-resistant ovarian cancer BC Breast cancer IIT Investigator initiated trial PVRIG Poliovirus receptor-related immunoglobulin BLA Biologics License Applications IL2 Interleukin 2 QxW Every x week(s) BMS Bristol Myers Squibb ILD Interstitial lung disease (ncc/cc)RCC ((non-)clear cell) Renal cell carcinoma CC Cervical cancer iNeST Individualized NeoAntigen-Specific Therapy SABCS San Antonio Breast Cancer Symposium CD-x Cluster of differentiation IO Immuno-oncology (ES/LS)SCLC (Extensive/low stage) small cell lung cancer CI Confidence interval JAMA Journal of the American Medical Association SD Stable disease CPI Checkpoint inhibitor JTO Journal of Thoracic Oncology SEC United States Securities and Exchange Commission CPS Combined positive score LALA IgG1 variant L234A/L235A SoC Standard of care CR Complete response MΦ Macrophage Teff Effector T cell CRC Colorectal cancer cMET Mesenchymal-Epithelial Transition factor TIGIT T cell immunoreceptor with Ig and ITIM domains (m)CRPC (metastatic) Castration resistant prostate cancer MIUC Muscle-invasive urothelial carcinoma TME Tumor microenvironment ctDNA Circulating tumor DNA MoA Mechanism of Action TNBC Triple-negative breast cancer CTLA Cytotoxic T-lymphocyte-associated protein MPM Malignant pleural mesothelioma T-Pam Trastuzumab pamirtecan DAR Drug-antibody ratio mRNA Messenger ribonucleic acid TPS Tumor proportion score DCR Disease control rate MSI-H High-frequence microsatellite instability TRAE Treatment-related adverse event EC Endometrial cancer MSS Microsatellite stability Treg Regulatory T cell EGFR Epidermal growth factor receptor NACLC North America Conference on Lung Cancer TROP2 Trophoblast cell-surface antigen 2 EpCAM Epithelial cell adhesion molecule NCT National clinical trial UC Urothelial cancer ESCC Esophageal squamous cell carcinoma NE Not evaluable for response VEGF(R) - A Vascular endothelial growth factor (receptor) A ESMO European Society for Medical Oncology NEN Neuroendocrine neoplasm VHH Heavy chain variable EU4(5) Includes Germany, France, Italy, Spain, UK NIH National Institutes of Health WBC White blood cell Fc Fragment crystallizable region NK cell Natural killer cell WCLC World Conference of Lung Cancer FDA Food and Drug Administration
